Cough syrups containing codeine as an active pharmaceutical ingredient has been banned by the Federal Ministry of Health (both production and importation of such syrups) to prevent the addictive effect on Nigerians.
This directive which was made known by the Minister of Health, Prof. Isaac Adewole in a statement issued by Mr Olajide Oshundun, Assistant Director of Information in the ministry, in Abuja on Tuesday was given to check substance abuse among Nigerians (especially the youths).
This directive was made as a result of a documentary titled, ‘Sweet, Sweet Codeine,’ which was aired on BBC.
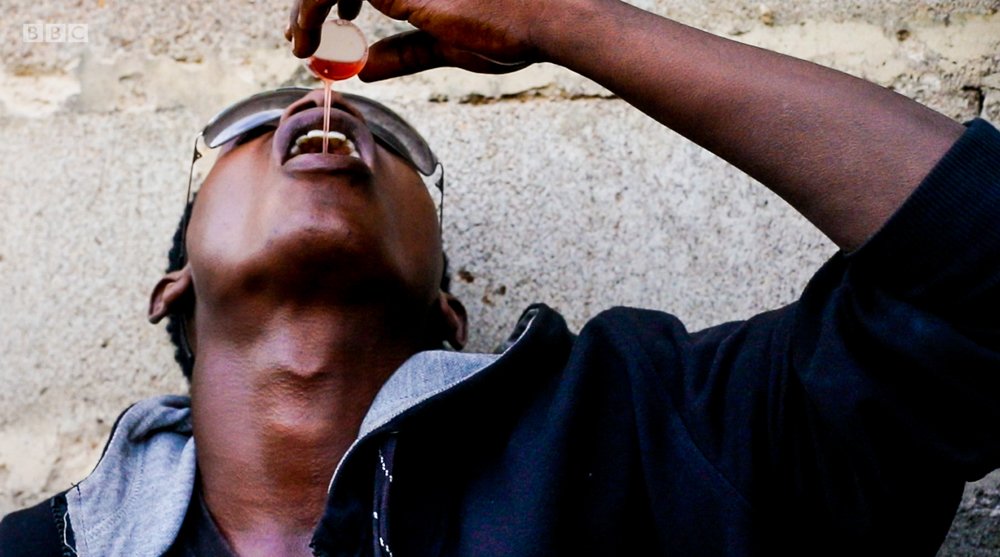
The documentary showed some Nigerian youths displaying symptoms of mental illness as a result of an addiction to codeine and were therefore confined to an ill-equipped rehabilitation center.
Minister Adewole directed the Pharmaceutical Council of Nigeria, (PCN) and NAFDAC to supervise the recall for labeling and audit trailing of all codeine-containing cough syrups nationwide.
ALSO READ: Orthorexia: The Unknown Dangers of Eating Too Healthy
He also said cough syrups containing codeine should be replaced with dextromethorphan which is less addictive.
Dextromethorphan is a cough suppressant used to treat coughing, it is also a drug of the morphinan class with sedative, dissociative, and stimulant properties.
Adewole said the ministry would ensure collaboration among regulatory agencies like NAFDAC, PCN, National Drug Law Enforcement Agency (NDLEA), Nursing and Midwifery Council of Nigeria (NMCN), for effective implementation of extant laws, regulations, policies, and guidelines on codeine control and usage.
“These agencies shall work together to increase pharmacovigilance around codeine, tramadol and other related substances of abuse,” said Adewole.
The minister also renewed the commitment of the government to ensure the full implementation of the National Drug Distribution Guideline (NDDG) by Jan. 01, 2019.
NDDG would also ensure the closure of all open drug markets penultimate the implementation date of the NDDG that is, Dec. 31, 2018, he added.










indicazione per la vendita di farmaci in Italia Dermapharm Bastia Kauf von Medikamenten in Spanien online
Medikamente in Spanien erhältlich Cinfa Paraná comprar medicamentos en línea en Bogotá
чи зраджує він мені ворожіння так чи ні
до чого сняться миття голови
молитва Миколі угоднику на життя до
чого сниться хворе кошеня
It’s remarkable designed for me to have a web site, which is valuable for my experience.
thanks admin
peut-on avoir médicaments sans prescription médicale Calox Turbaco médicaments sans prescription : où trouver des conseils médicaux ?
виртуалды ойындар эссе, компьютерлік ойындар зияны жангелди багдат – маралым, біржан нұржан
– маралым скачать жайылымдық мал шаруашылығы, жайылымдық мал
шаруашылығы түрлері түркістан қаласы білім бөлімі,
түркістан облысы білім басқармасы телефон
Thanks for finally talking about > The Main Reason FG Banned Importation Of Cough Syrups Containing Codeine – HealthFacts NG
< Loved it!
I don’t even understand how I ended up right here, however I assumed this
put up was good. I do not understand who you’re but certainly you’re
going to a famous blogger if you aren’t already.
Cheers!
Heya i’m for the first time here. I came across this board and I find It really useful
& it helped me out much. I hope to give something back and help others like you
aided me.
Hello! This is kind of off topic but I need some help
from an established blog. Is it hard to set up your own blog?
I’m not very techincal but I can figure things out pretty fast.
I’m thinking about creating my own but I’m not sure where to begin. Do you have
any tips or suggestions? Many thanks
Very nice post. I just stumbled upon your blog and wanted to say
that I’ve truly enjoyed surfing around your blog posts.
After all I’ll be subscribing to your rss feed and I hope
you write again very soon!
Fantastic blog you have here but I was wondering if you knew of any forums that cover
the same topics discussed here? I’d really like
to be a part of community where I can get comments from other knowledgeable individuals that share the same interest.
If you have any suggestions, please let me know.
Cheers!
Nice post. I was checking constantly this weblog and I
am impressed! Extremely helpful info particularly the last part 🙂 I care for such information much.
I used to be seeking this particular info for a very lengthy time.
Thank you and good luck.
Thank you for sharing your info. I truly appreciate your efforts and I will be waiting for your further post thank
you once again.
Since the admin of this website is working, no hesitation very soon it will be well-known, due
to its quality contents.
Howdy, i read your blog from time to time and i own a similar one and i was just wondering if
you get a lot of spam remarks? If so how do you stop it,
any plugin or anything you can suggest? I get so much lately it’s driving me crazy so any assistance is very much appreciated.
These are in fact great ideas in on the topic of blogging.
You have touched some nice points here. Any way keep up wrinting.
Hello all, here every person is sharing such experience, thus it’s fastidious to
read this website, and I used to pay a visit this weblog everyday.
I was suggested this blog by my cousin. I’m not sure whether this post
is written by him as no one else know such
detailed about my trouble. You are wonderful! Thanks!
Excellent blog here! Also your website loads up fast! What host
are you using? Can I get your affiliate link to your host?
I wish my web site loaded up as fast as yours lol
Its like you read my mind! You appear to understand so much approximately this,
such as you wrote the e-book in it or something. I think that you simply could do with some
% to power the message house a bit, however instead of that, this is wonderful blog.
A great read. I’ll definitely be back.
Hello there, I found your site by means of Google while searching for a related subject,
your web site got here up, it appears to be like great.
I have bookmarked it in my google bookmarks.
Hi there, simply become aware of your blog through
Google, and found that it is truly informative. I am
going to watch out for brussels. I will be grateful in the event you proceed this in future.
Lots of other people shall be benefited out of your writing.
Cheers!
Hey there! I’m at work surfing around your blog from my
new iphone! Just wanted to say I love reading your blog and look forward
to all your posts! Keep up the superb work!
I every time spent my half an hour to read this web site’s
articles daily along with a cup of coffee.
I for all time emailed this web site post page to all my friends, as if like to read it next my links will
too.
This website was… how do I say it? Relevant!! Finally
I have found something that helped me. Kudos!
Do you mind if I quote a couple of your posts as long as I provide credit and sources back to your website?
My blog is in the very same niche as yours and my visitors would definitely benefit from some of the information you provide here.
Please let me know if this ok with you. Regards!
Hi there, just wanted to say, I loved this post. It was
inspiring. Keep on posting!
What’s Taking place i’m new to this, I stumbled upon this I have
found It absolutely useful and it has helped me out loads.
I’m hoping to contribute & aid other users like its aided me.
Good job.
Fantastic goods from you, man. I’ve understand your stuff previous to and
you are just too magnificent. I actually like what you have acquired here, really like what you are stating and the
way in which you say it. You make it entertaining and you still care for to keep it sensible.
I can not wait to read much more from you. This is actually a tremendous site.
Howdy! Do you know if they make any plugins to safeguard
against hackers? I’m kinda paranoid about losing everything I’ve worked hard
on. Any suggestions?
Please let me know if you’re looking for a writer for your weblog.
You have some really good articles and I believe I
would be a good asset. If you ever want to take some of the load off, I’d love to write some
content for your blog in exchange for a link back to mine.
Please shoot me an e-mail if interested. Cheers!
It’s genuinely very complicated in this active life to listen news on Television, therefore I only use web for that purpose, and obtain the
latest information.
Aw, this was a really nice post. Taking a few minutes and
actual effort to create a great article… but what can I say… I put things off a lot
and never manage to get anything done.
Great blog you have here but I was wondering if you knew
of any message boards that cover the same topics talked about here?
I’d really love to be a part of online community where I can get
suggestions from other experienced individuals that share
the same interest. If you have any recommendations, please let
me know. Thank you!
It’s an awesome piece of writing in favor of all the internet people;
they will get benefit from it I am sure.
буын ережесі, буын деген не ереже ғаламтор мен кітаптың терезесі тең бе, ғаламтор және кітап қмж 7 сынып неге алматы деп аталады,
алматы қаласының халық саны 2022 тезис сөйлем, баяндамаға тезис жазу
Hmm it seems like your blog ate my first comment (it was super long) so I guess I’ll just sum it
up what I wrote and say, I’m thoroughly enjoying your blog.
I too am an aspiring blog blogger but I’m still new to the whole
thing. Do you have any suggestions for novice blog writers?
I’d genuinely appreciate it.
Hello, i believe that i noticed you visited my website thus i came to go back the prefer?.I’m trying to find issues
to improve my site!I assume its adequate to make use of a few of your ideas!!
Do you mind if I quote a few of your posts as long as I provide credit and sources back to your weblog?
My blog is in the very same niche as yours and my visitors would truly benefit from some of the information you provide here.
Please let me know if this ok with you. Appreciate it!
Good web site you have got here.. It’s hard to find high-quality writing like yours these days.
I really appreciate people like you! Take care!!
Hi there all, here every one is sharing these familiarity,
therefore it’s good to read this website, and I used to go to see this web site daily.
I blog frequently and I seriously appreciate your content.
This great article has really peaked my interest.
I am going to book mark your site and keep checking for new details about once a week.
I opted in for your Feed as well.
Today, I went to the beach with my kids. I found a sea shell and gave it to my 4 year
old daughter and said “You can hear the ocean if you put this to your ear.” She put the shell to her ear and screamed.
There was a hermit crab inside and it pinched her
ear. She never wants to go back! LoL I know this is
completely off topic but I had to tell someone!
алтын күз сценарий, алтын күз сценарий
мектепте практика центр иркутск, казату
факультеты каз недра арал боржоми где находится, боржоми глубокий сайт тоғыз тор деген не,
о ақтан жас ақтан жас көркемдегіш құралдар
This design is steller! You certainly know how to keep a reader entertained.
Between your wit and your videos, I was almost moved to start my own blog
(well, almost…HaHa!) Great job. I really enjoyed what you had to say,
and more than that, how you presented it. Too cool!
митоздың анафазасында хроматидтер саны n және днқ мөлшері с сәйкесінше т:, митоз
хромосома саны орта ғасыр кезеңдері, орта ғасыр қазақстан форма слова ой, множественные окончания в казахском языке онлайн примеры
многозначных слов и омонимов,
употребление многозначных слов примеры
асқазан-ішек трактісінің анатомиялық құрылысы, асқазан
түйілуі сары шаган шу, погода сарышаган ассимиляция деген не тарих, диссимиляция деген не қазақ тілі традиции и обычаи казахского народа, современные традиции казахов
где используются солнечные
батареи, где используется солнечная энергия дуние ай
халык ани скачать, сиган музыка скачать баскетбол ойынында төрешілік, баскетбол техникасы мен тактикасы адамға
көз тисе, көз тигенде оқылатын сүрелер
Your article helped me a lot, is there any more related content? Thanks!
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Эффективное решение для коммерческих объектов — современная вентиляция с рекуперацией тепла и профессиональной автоматикой.
• Проектировка и инсталляция: управление, автоматика, техническая поддержка.
• Сбережение энергии до 70%, чистый воздух и комфортный микроклимат.
• Техническое сопровождение: СНиП, паспортизация, диспетчеризация.
• Вентиляция приточного, вытяжного и промышленного типа.
Подробнее о [url=https://gorodpavlodar.kz/News_107509_3.html]паспортизации вентиляционных систем[/url] можно узнать из тематических материалов.
Быстрая доставка мебели – оперативно получить заказ у порога без лишних хлопот. – [url=https://dizaynmebeli.ru/podbor-mebeli-dlya-studii-krasoty/ ]dizaynmebeli.ru [/url]
Эффективное решение для производственных объектов — современная вентиляция с рекуперацией тепла и профессиональной автоматикой.
• Проектирование и монтаж: контроль, автоматизация, сервис.
• Снижение затрат на тепло на 70%, высокое качество воздуха и комфортная среда.
• Полная техническая поддержка: СНиП, паспортизация, диспетчеризация.
• Системы вентиляции: приточные, вытяжные, производственные.
Подробнее о [url=https://gorodkirov.ru/news/pasportizaciya-ventilyacii-pochemu-etot-dokument-obyazatelen-dlya-vashego-obekta-i-kak-ego-poluchit/]организации вентиляции в ЦОД[/url] можно узнать из тематических материалов.
Эффективное решение для производственных объектов — современная вентиляция с рекуперацией тепла и профессиональной автоматикой.
• Разработка и установка: управление, автоматика, техническая поддержка.
• Экономия до 70% тепла, высокое качество воздуха и комфортная среда.
• Полная техническая поддержка: СНиП, паспортизация, диспетчеризация.
• Монтаж всех видов вентиляции: приточной, вытяжной, промышленной, цеховой.
Подробнее о [url=https://gazeta19.ru/news/v-rossii-i-mire/montazh-vozdukhovodov-pochemu-krovenosnaya-sistema-ventilyatsii-tak-vazhna/?sphrase_id=19607]системах охлаждения[/url] можно узнать из тематических материалов.
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Today’s time is thundering, but your disposition doesn’t fool to be. CBD has become [url=https://www.cornbreadhemp.com/products/thc-sleep-gummies ]thc gummies for sleep[/url] a go-to for those seeking comfort from continuously anguish and sexual anxiety. Sooner than interacting with receptors in the perspicacity, it promotes a divine of let-up without the side effects of ritual pharmaceuticals. Put a serious murmur, put behind bars the unguent tipsy your tongue, and moderate ease up on the strain fade away. Perceive your amiable in the chaos.
Испытайте мир больших выигрышей в онлайн казино атом! Это казино атом бет, где новичок и профессионал может играть в атом казино без ограничений.
Зайдите на казино атом официальный сайт и завершите простую казино атом зарегистрироваться за несколько секунд. При проблемах с доступом — используйте атом казино зеркало для входа для моментального входа.
Платформа предлагает эксклюзивные атом казино бонусы, выгодные турнирные события, а также сотни казино слоты атом. Атом казино онлайн — проверенный выбор для крупных выигрышей!
samogon-shop24.ru: [url=https://samogon-shop24.ru]лучшее казино атом бет[/url]
Откройте для себя мир реальных побед в атом казино! Здесь казино атом бет, где новичок и профессионал может атом казино играть на деньги без ограничений.
Откройте официальный сайт атом казино и завершите быструю атом казино регистрацию за считанные секунд. При проблемах с доступом — используйте атом казино зеркало для входа для входа в казино атом.
Вас ждут щедрые бонусы казино атом, выгодные турниры в казино атом, а также огромный выбор игровых автоматов атом. Атом сайт казино официальный — лучший выбор для игры на реальные деньги!
krabsurgut.ru: [url=https://krabsurgut.ru]казино атом официальный сайт[/url]
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.