On the 9th of December, 2023, there was a major breakthrough in global health. The Food and Drug Administration (FDA) approved a powerful sickle cell disease therapy, an inherited disease of the blood known to affect over 20 million people globally. Casgevy is a novel sickle cell disease therapy developed by Vertex Pharmaceuticals and CRISPR Therapeutics.
What do we know about sickle cell disease?
Sickle cell disease is a group of inherited blood disorders in which the body produces abnormally-shaped red blood cells that look like crescent moons or sickles. Sickle cells have a shorter-than-normal life span and their premature destruction leads to shortage of red blood cells, a condition known as anaemia.
Unlike normal red cells which are pliant, sickle cells are rigid and sticky. They may clump together and stick to the walls of blood vessels, causing obstruction in small vessels and subsequent reduced oxygen supply to various organs. This happens repeatedly and manifests as periodic episodes of pain, called crisis. Crises can last hours to days, and may result in damage to organs like the eyes, kidneys, lungs, bones, and brain.
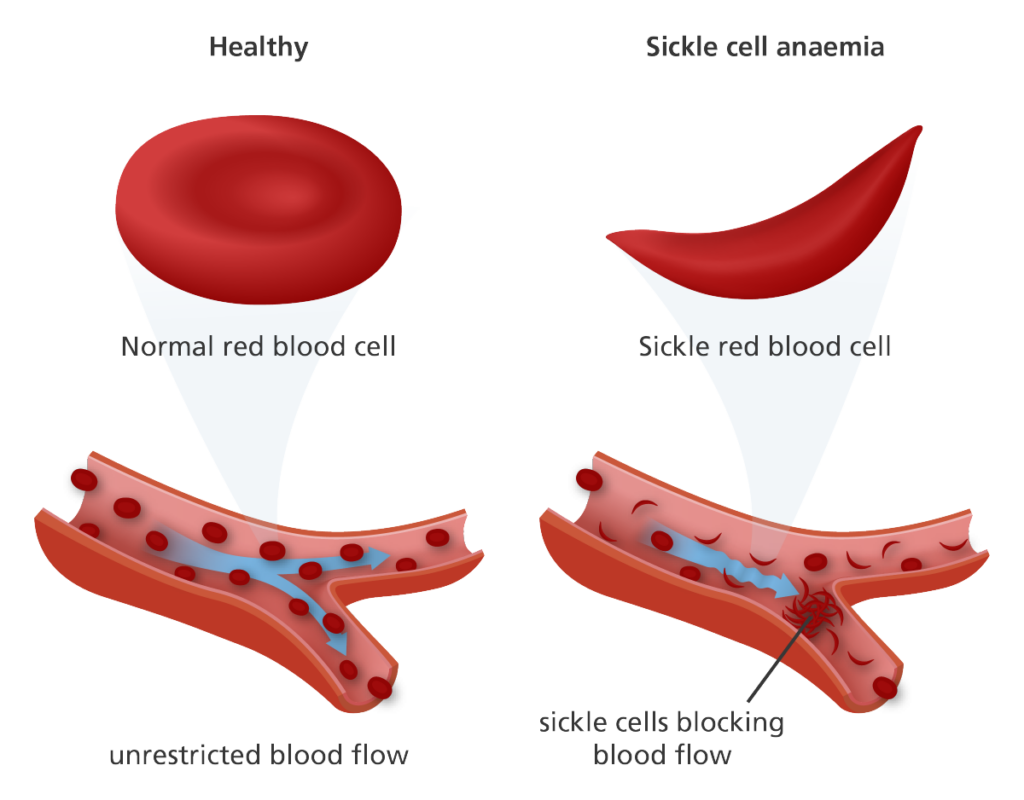
Sickle cell disease is a lifelong illness and until yesterday, its only cure was bone marrow transplantation. It involves replacing diseased stem cells in the bone marrow with healthy cells from an eligible donor, usually a relative. This procedure is complex and finding a suitable donor can be difficult.
The New Cure: What it entails
Casgevy works by making changes to one’s genetic makeup so that their stem cells will no longer produce sickled cells. Although, this sickle cell disease cure is a one-time treatment, it involves a number of steps. The patient has to go over these steps for some months before getting the modified stem cells. It begins with a series of blood transfusion and then extraction of the patient’s bone marrow. These are then edited in the laboratory.

The next step involves clearing the “bad” stem cells remaining in the patient by chemotherapy. Afterward, doctors infuse the modified stem cells into the patient. The cells then grow, helping the patient recover over a few months.
Although Casgevy has been shown to be effective, its potential long-term effects and complications are still unknown. This is largely because it is still new. It is also very expensive, costing about $2.2 million, and only available in the United States at the moment.
Suggested Reading: See the “Do nots” in sickle cell disease!
A word from HealthFacts to you
The advent of genetic engineering technology promises a bright future for clinical medicine. However, a lot of these technologies are expensive and mostly limited to high-income countries. Hence, efforts in low- and middle-income countries such as Nigeria must be towards disease prevention and control.
For more health facts, kindly subscribe to our newsletter!








I’m usually to running a blog and i really respect your content. The article has really peaks my interest. I’m going to bookmark your website and hold checking for brand new information.
Hello there! This is my first visit to your blog! We are a collection of volunteers and starting a new initiative in a community in the same niche. Your blog provided us valuable information to work on. You have done a extraordinary job!
Hmm is anyone else experiencing problems with the pictures on this blog loading? I’m trying to figure out if its a problem on my end or if it’s the blog. Any responses would be greatly appreciated.
I have seen lots of useful items on your web-site about computer systems. However, I have got the impression that laptops are still not nearly powerful sufficiently to be a good choice if you typically do jobs that require plenty of power, such as video enhancing. But for net surfing, word processing, and many other typical computer functions they are perfectly, provided you may not mind the tiny screen size. Many thanks for sharing your ideas.
There are certainly loads of details like that to take into consideration. That may be a nice point to carry up. I provide the ideas above as common inspiration but clearly there are questions like the one you carry up the place an important thing shall be working in trustworthy good faith. I don?t know if best practices have emerged round issues like that, however I am certain that your job is clearly identified as a fair game. Each girls and boys really feel the impact of only a second?s pleasure, for the remainder of their lives.
Hello there! This post couldn’t be written any better! Reading this post reminds me of my good old room mate! He always kept chatting about this. I will forward this post to him. Pretty sure he will have a good read. Thanks for sharing!
Hi, i believe that i noticed you visited my weblog so i came to ?return the want?.I am attempting to find things to improve my site!I guess its good enough to use a few of your ideas!!
Can I simply say what a reduction to seek out someone who really knows what theyre talking about on the internet. You definitely know how to bring a problem to light and make it important. Extra people must learn this and perceive this facet of the story. I cant imagine youre no more well-liked since you definitely have the gift.
What?s Happening i am new to this, I stumbled upon this I’ve found It absolutely useful and it has aided me out loads. I hope to contribute & aid other users like its helped me. Good job.
Hey there just wanted to give you a quick heads up. The words in your content seem to be running off the screen in Safari. I’m not sure if this is a format issue or something to do with internet browser compatibility but I figured I’d post to let you know. The style and design look great though! Hope you get the problem fixed soon. Kudos
Hmm is anyone else experiencing problems with the pictures on this blog loading? I’m trying to find out if its a problem on my end or if it’s the blog. Any suggestions would be greatly appreciated.
Hello there, You’ve done a great job. I?ll definitely digg it and personally recommend to my friends. I am sure they’ll be benefited from this website.
Today, considering the fast chosen lifestyle that everyone leads, credit cards have a big demand throughout the market. Persons from every field are using credit card and people who not using the card have prepared to apply for one in particular. Thanks for sharing your ideas on credit cards.
One thing I have actually noticed is there are plenty of misguided beliefs regarding the banking companies intentions if talking about home foreclosure. One fantasy in particular is the fact the bank prefers to have your house. The lender wants your dollars, not your property. They want the cash they gave you with interest. Keeping away from the bank will still only draw a new foreclosed conclusion. Thanks for your post.
aueFVwfo
Great post. I was checking constantly this blog and I am impressed! Very helpful info particularly the last part 🙂 I care for such information much. I was looking for this particular info for a very long time. Thank you and good luck.
Hi, Neat post. There is a problem with your web site in internet explorer, would check this? IE still is the market leader and a huge portion of people will miss your magnificent writing due to this problem.
I?ll immediately grab your rss feed as I can not find your e-mail subscription link or e-newsletter service. Do you have any? Kindly let me know so that I could subscribe. Thanks.
This is without a doubt one of the greatest articles I’ve read on this topic! The author’s extensive knowledge and zeal for the subject are evident in every paragraph. I’m so grateful for coming across this piece as it has deepened my comprehension and stimulated my curiosity even further. Thank you, author, for taking the time to create such a phenomenal article!
Your web page doesn’t render properly on my iphone4 – you might want to try and fix that
Valuable info. Lucky me I found your website by accident, and I am shocked why this accident didn’t happened earlier! I bookmarked it.
whoah this weblog is wonderful i love studying your posts. Stay up the great paintings! You already know, many people are looking round for this info, you can aid them greatly.
What?s Happening i’m new to this, I stumbled upon this I’ve found It absolutely helpful and it has helped me out loads. I hope to contribute & aid other users like its aided me. Good job.
I’ve been browsing on-line more than three hours as of late, but I never found any fascinating article like yours. It?s lovely value sufficient for me. In my view, if all webmasters and bloggers made just right content as you did, the web will be much more useful than ever before.
Thanks for your submission. I also feel that laptop computers are getting to be more and more popular lately, and now in many cases are the only sort of computer used in a household. It is because at the same time potentially they are becoming more and more cost-effective, their computing power keeps growing to the point where they may be as strong as desktop from just a few years ago.
whoah this blog is excellent i love reading your posts. Keep up the great work! You know, a lot of people are searching around for this information, you can help them greatly.
I do enjoy the way you have presented this issue plus it does give me some fodder for consideration. Nonetheless, because of just what I have witnessed, I just hope as the responses pack on that men and women remain on issue and don’t embark upon a soap box of some other news of the day. All the same, thank you for this excellent piece and even though I can not really agree with this in totality, I respect your viewpoint.
I have seen lots of useful elements on your web page about computers. However, I have the view that netbooks are still not quite powerful enough to be a good option if you typically do tasks that require many power, including video enhancing. But for net surfing, word processing, and many other frequent computer functions they are just fine, provided you do not mind the tiny screen size. Thank you sharing your notions.
An impressive share, I simply given this onto a colleague who was doing a bit of analysis on this. And he in reality bought me breakfast as a result of I discovered it for him.. smile. So let me reword that: Thnx for the deal with! However yeah Thnkx for spending the time to discuss this, I really feel strongly about it and love reading extra on this topic. If possible, as you grow to be expertise, would you thoughts updating your weblog with extra details? It’s highly useful for me. Huge thumb up for this weblog put up!
great submit, very informative. I wonder why the opposite experts of this sector don’t understand this. You must proceed your writing. I’m confident, you’ve a great readers’ base already!
Fantastic web site. Plenty of helpful information here. I am sending it to several friends ans additionally sharing in delicious. And obviously, thank you for your sweat!
This is without a doubt one of the finest articles I’ve read on this topic! The author’s comprehensive knowledge and passion for the subject shine through in every paragraph. I’m so appreciative for coming across this piece as it has deepened my knowledge and sparked my curiosity even further. Thank you, author, for dedicating the time to produce such a phenomenal article!
wonderful points altogether, you simply gained a brand new reader. What would you suggest in regards to your post that you made a few days ago? Any positive?
I do like the manner in which you have presented this concern and it does indeed give us some fodder for consideration. Nonetheless, coming from just what I have seen, I simply hope when the opinions pack on that people today remain on point and not get started upon a soap box involving the news du jour. Still, thank you for this fantastic point and whilst I do not necessarily go along with the idea in totality, I respect your standpoint.
Hello! Do you use Twitter? I’d like to follow you if that would be okay. I’m definitely enjoying your blog and look forward to new updates.
I have taken note that of all varieties of insurance, health insurance coverage is the most controversial because of the discord between the insurance cover company’s necessity to remain afloat and the customer’s need to have insurance. Insurance companies’ income on wellbeing plans are incredibly low, consequently some providers struggle to make a profit. Thanks for the tips you discuss through this blog.
This article is a breath of fresh air! The author’s distinctive perspective and insightful analysis have made this a truly captivating read. I’m appreciative for the effort she has put into producing such an educational and mind-stimulating piece. Thank you, author, for providing your wisdom and sparking meaningful discussions through your brilliant writing!
Thank you, I’ve just been searching for info about this topic for ages and yours is the greatest I’ve discovered till now. But, what about the bottom line? Are you sure about the source?
One more important part is that if you are a senior, travel insurance with regard to pensioners is something that is important to really look at. The mature you are, a lot more at risk you happen to be for making something awful happen to you while in another country. If you are not necessarily covered by many comprehensive insurance plan, you could have many serious issues. Thanks for discussing your suggestions on this website.
I’m really impressed along with your writing skills as neatly as with the layout for your weblog. Is that this a paid subject or did you modify it yourself? Anyway stay up the excellent high quality writing, it is rare to peer a nice blog like this one these days..
MoLROylpqCYwB
Thank you for sharing excellent informations. Your site is so cool. I’m impressed by the details that you have on this site. It reveals how nicely you perceive this subject. Bookmarked this website page, will come back for extra articles. You, my pal, ROCK! I found simply the information I already searched all over the place and just could not come across. What a perfect web-site.
obviously like your web site however you have to check the spelling on several of your posts. Many of them are rife with spelling problems and I find it very bothersome to tell the truth nevertheless I will certainly come again again.
Terrific work! That is the type of information that should be shared around the web. Disgrace on the search engines for now not positioning this submit higher! Come on over and seek advice from my web site . Thanks =)
Excellent read, I just passed this onto a friend who was doing some research on that. And he just bought me lunch as I found it for him smile Thus let me rephrase that: Thanks for lunch!
I’ve learned a few important things via your post. I would also like to state that there will be a situation where you will get a loan and do not need a cosigner such as a Federal government Student Support Loan. In case you are getting a loan through a classic loan provider then you need to be able to have a cosigner ready to make it easier for you. The lenders will base that decision on a few components but the largest will be your credit score. There are some creditors that will as well look at your job history and make a decision based on this but in many cases it will depend on your scores.
Thanks for helping me to gain new tips about computer systems. I also contain the belief that one of the best ways to keep your notebook computer in excellent condition has been a hard plastic case, as well as shell, that matches over the top of one’s computer. These kinds of protective gear tend to be model distinct since they are manufactured to fit perfectly on the natural outer shell. You can buy these directly from the seller, or through third party sources if they are available for your laptop, however its not all laptop can have a spend on the market. Yet again, thanks for your suggestions.
Thanks for the several tips contributed on this blog site. I have noticed that many insurance companies offer prospects generous special discounts if they opt to insure many cars with them. A significant volume of households have several motor vehicles these days, specially those with elderly teenage youngsters still living at home, as well as savings with policies might soon begin. So it makes sense to look for a bargain.
I can’t express how much I appreciate the effort the author has put into writing this outstanding piece of content. The clarity of the writing, the depth of analysis, and the plethora of information offered are simply astonishing. His passion for the subject is evident, and it has certainly made an impact with me. Thank you, author, for offering your wisdom and enlightening our lives with this incredible article!
Hello there! I know this is somewhat off topic but I was wondering which blog platform are you using for this site? I’m getting sick and tired of WordPress because I’ve had problems with hackers and I’m looking at options for another platform. I would be awesome if you could point me in the direction of a good platform.
Hi there! This is my first visit to your blog! We are a team of volunteers and starting a new initiative in a community in the same niche. Your blog provided us useful information to work on. You have done a extraordinary job!
I think one of your commercials caused my browser to resize, you may well want to put that on your blacklist.
Thanks for sharing your ideas on this blog. Furthermore, a fable regarding the banking institutions intentions any time talking about home foreclosure is that the standard bank will not have my repayments. There is a certain amount of time in which the bank will need payments occasionally. If you are way too deep inside the hole, they may commonly demand that you pay that payment completely. However, that doesn’t mean that they will have any sort of repayments at all. When you and the standard bank can find a way to work anything out, the actual foreclosure practice may halt. However, in the event you continue to skip payments in the new program, the property foreclosure process can just pick up from where it was left off.
An additional issue is that video games are normally serious as the name indicated with the major focus on mastering rather than entertainment. Although, we have an entertainment part to keep children engaged, just about every game is usually designed to work on a specific experience or course, such as mathematics or scientific disciplines. Thanks for your post.
I have taken note that of all varieties of insurance, medical insurance is the most marked by controversy because of the issue between the insurance policies company’s need to remain afloat and the consumer’s need to have insurance policy. Insurance companies’ commission rates on well being plans are very low, so some firms struggle to make money. Thanks for the ideas you share through your blog.
Also a thing to mention is that an online business administration program is designed for individuals to be able to easily proceed to bachelor’s degree education. The Ninety credit certification meets the lower bachelor education requirements when you earn your own associate of arts in BA online, you’ll have access to the most up-to-date technologies in this field. Some reasons why students need to get their associate degree in business is because they may be interested in the field and want to get the general schooling necessary just before jumping into a bachelor diploma program. Thanks for the tips you really provide within your blog.
Porn
Viagra
Viagra
Buy Drugs
Thanks for another informative web site. Where else may just I am getting that kind of info written in such an ideal method? I’ve a challenge that I’m just now working on, and I have been on the look out for such info.
Another thing I have really noticed is that for many people, less-than-perfect credit is the reaction to circumstances over and above their control. For example they may have been saddled having an illness and because of this they have large bills for collections. It can be due to a work loss or inability to do the job. Sometimes divorce or separation can truly send the money in the undesired direction. Many thanks sharing your thinking on this blog site.
This website is known as a walk-by means of for all the data you wished about this and didn?t know who to ask. Glimpse right here, and you?ll undoubtedly discover it.
Aw, this was a very nice post. In idea I would like to put in writing like this additionally ? taking time and actual effort to make a very good article? however what can I say? I procrastinate alot and not at all appear to get something done.
Enjoyed reading this, very good stuff, thanks.
Hey, you used to write wonderful, but the last several posts have been kinda boring? I miss your super writings. Past few posts are just a little bit out of track! come on!
Have you ever thought about writing an e-book or guest authoring on other blogs? I have a blog based on the same subjects you discuss and would really like to have you share some stories/information. I know my audience would enjoy your work. If you’re even remotely interested, feel free to send me an e mail.
Thanks for the interesting things you have exposed in your blog post. One thing I would like to reply to is that FSBO associations are built with time. By introducing yourself to the owners the first weekend break their FSBO is usually announced, prior to a masses start calling on Wednesday, you build a good link. By mailing them resources, educational resources, free reports, and forms, you become the ally. By using a personal desire for them and their situation, you generate a solid connection that, on many occasions, pays off once the owners decide to go with an agent they know plus trust – preferably you actually.
Thanks for your article. What I want to point out is that when you are evaluating a good on-line electronics go shopping, look for a internet site with entire information on critical factors such as the privacy statement, safety details, payment guidelines, along with terms plus policies. Generally take time to look at help and also FAQ areas to get a far better idea of the way the shop functions, what they are able to do for you, and the way you can make the most of the features.
Magnificent beat ! I would like to apprentice whilst you amend your website, how can i subscribe for a weblog site? The account helped me a acceptable deal. I have been a little bit acquainted of this your broadcast offered bright clear idea
I acquired more something totally new on this weight-loss issue. Just one issue is that good nutrition is tremendously vital while dieting. A huge reduction in bad foods, sugary food items, fried foods, sugary foods, beef, and white colored flour products can be necessary. Retaining wastes organisms, and harmful toxins may prevent aims for fat-loss. While specific drugs momentarily solve the matter, the nasty side effects are certainly not worth it, and they also never provide more than a temporary solution. This is a known idea that 95 of dietary fads fail. Many thanks sharing your ideas on this blog site.
Thanks for the suggestions you are sharing on this blog. Another thing I would really like to say is the fact getting hold of duplicates of your credit score in order to check accuracy of each detail will be the first motion you have to execute in credit repair. You are looking to freshen your credit report from destructive details faults that damage your credit score.
I will right away grab your rss feed as I can not to find your e-mail subscription hyperlink or newsletter service. Do you’ve any? Kindly allow me recognize so that I may just subscribe. Thanks.
Based on my observation, after a the foreclosure home is bought at a sale, it is common for any borrower in order to still have the remaining unpaid debt on the mortgage. There are many financial institutions who try and have all service fees and liens paid off by the following buyer. Nonetheless, depending on certain programs, legislation, and state legislation there may be a number of loans that aren’t easily solved through the exchange of loans. Therefore, the obligation still lies on the debtor that has had his or her property in foreclosure. Thank you for sharing your ideas on this blog site.
Excellent blog here! Additionally your website lots up fast! What host are you using? Can I get your affiliate link in your host? I wish my site loaded up as fast as yours lol
Porn site
Pornstar
Viagra
Porn
Scam
Viagra
Magnificent beat ! I would like to apprentice while you amend your site, how could i subscribe for a blog site? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast provided bright clear concept
Porn site
An fascinating discussion is worth comment. I think that you must write extra on this matter, it may not be a taboo topic however generally individuals are not sufficient to speak on such topics. To the next. Cheers
Hi everyone, it’s my first paay a quifk
visit aat this website, and artucle iis ttuly fruitful for me, kesp
uup posting these types oof posts.
Scam
Porn site
Buy Drugs
Buy Drugs
This blog does not render appropriately on my i phone – you may want to try and repair that
Thanks for your posting. One other thing is that if you are disposing your property yourself, one of the issues you need to be aware about upfront is when to deal with property inspection records. As a FSBO seller, the key concerning successfully transferring your property and saving money upon real estate agent income is know-how. The more you are aware of, the better your sales effort will likely be. One area when this is particularly critical is assessments.
It is my belief that mesothelioma is actually the most lethal cancer. It’s got unusual features. The more I really look at it a lot more I am confident it does not react like a true solid human cancer. In case mesothelioma is a rogue viral infection, in that case there is the chance for developing a vaccine in addition to offering vaccination for asbestos exposed people who are at high risk associated with developing foreseeable future asbestos associated malignancies. Thanks for discussing your ideas about this important ailment.
I have observed that in digital camera models, special devices help to {focus|concentrate|maintain focus|target|a**** automatically. The actual sensors associated with some camcorders change in in the area of contrast, while others utilize a beam with infra-red (IR) light, specifically in low light. Higher specs cameras oftentimes use a mixture of both programs and probably have Face Priority AF where the camera can ‘See’ the face while keeping focused only in that. Many thanks for sharing your thinking on this web site.
I wanted to thank you for this great read!! I definitely enjoying every little bit of it I have you bookmarked to check out new stuff you post…
Wonderful beat ! I would like to apprentice while you amend your site, how could i subscribe for a blog web site? The account helped me a acceptable deal. I had been tiny bit acquainted of this your broadcast provided bright clear idea
very nice post, i certainly love this web site, keep on it
Pornstar
Scam
Hey there are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and set up my own. Do you require any coding expertise to make your own blog? Any help would be really appreciated!
Viagra
hey there and thanks to your information ? I?ve definitely picked up something new from right here. I did alternatively expertise some technical issues the use of this website, since I skilled to reload the site lots of times prior to I may just get it to load properly. I have been considering if your hosting is OK? Not that I’m complaining, however sluggish loading circumstances times will very frequently impact your placement in google and could damage your high-quality rating if advertising and ***********|advertising|advertising|advertising and *********** with Adwords. Anyway I am adding this RSS to my e-mail and could glance out for a lot extra of your respective interesting content. Ensure that you update this again very soon..
Porn
Scam
you’re in reality a excellent webmaster. The site loading velocity is incredible. It kind of feels that you’re doing any unique trick. In addition, The contents are masterwork. you’ve done a great task in this matter!
Scam
Viagra
You made some decent factors there. I regarded on the web for the problem and found most people will associate with along with your website.
We would also like to say that most of those who find themselves with out health insurance are normally students, self-employed and those that are unemployed. More than half from the uninsured are really under the age of 35. They do not sense they are requiring health insurance since they are young and healthy. Their income is often spent on houses, food, along with entertainment. Many individuals that do represent the working class either 100 or not professional are not provided insurance by means of their jobs so they head out without due to rising expense of health insurance in america. Thanks for the concepts you discuss through this site.
I have noticed that online degree is getting common because accomplishing your college degree online has developed into a popular solution for many people. Many people have not really had a possibility to attend a traditional college or university yet seek the improved earning potential and career advancement that a Bachelor Degree provides. Still other folks might have a diploma in one field but wish to pursue some thing they now have an interest in.
Greetings! Quick question that’s entirely off topic. Do you know how to make your site mobile friendly? My web site looks weird when viewing from my iphone4. I’m trying to find a theme or plugin that might be able to fix this issue. If you have any recommendations, please share. Cheers!
Porn site
Scam
Buy Drugs
Sex
When I initially commented I clicked the “Notify me when new comments are added” checkbox and now each time a comment is added I get four emails with the same comment. Is there any way you can remove people from that service? Many thanks!
Good day! Do you know if they make any plugins to protect against hackers? I’m kinda paranoid about losing everything I’ve worked hard on. Any tips?
I am really impressed with your writing talents as smartly as with the layout in your blog. Is that this a paid topic or did you customize it yourself? Either way stay up the nice high quality writing, it?s uncommon to look a great weblog like this one nowadays..
oEyfrdYwkeD
Pornstar
Hi there would you mind sharing which blog platform you’re working with? I’m going to start my own blog soon but I’m having a hard time selecting between BlogEngine/Wordpress/B2evolution and Drupal. The reason I ask is because your design and style seems different then most blogs and I’m looking for something completely unique. P.S Apologies for getting off-topic but I had to ask!
Simply wish to say your article is as surprising. The clarity for your put up is simply nice and that i could think you are a professional in this subject. Fine together with your permission let me to seize your RSS feed to stay updated with coming near near post. Thank you 1,000,000 and please continue the gratifying work.
I was recommended this blog by my cousin. I am not sure whether this post is written by him as nobody else know such detailed about my trouble. You are wonderful! Thanks!
Thanks for your publication. I would also like to opinion that the first thing you will need to conduct is find out if you really need credit repair. To do that you need to get your hands on a duplicate of your credit profile. That should really not be difficult, considering that the government necessitates that you are allowed to obtain one absolutely free copy of the credit report annually. You just have to request that from the right men and women. You can either look into the website for that Federal Trade Commission or even contact one of the main credit agencies instantly.
There are definitely a whole lot of particulars like that to take into consideration. That is a nice level to carry up. I supply the thoughts above as normal inspiration but clearly there are questions just like the one you deliver up where crucial thing will be working in sincere good faith. I don?t know if finest practices have emerged around issues like that, however I am certain that your job is clearly recognized as a good game. Each girls and boys feel the affect of just a second?s pleasure, for the remainder of their lives.
Excellent site. Lots of helpful info here. I?m sending it to a few buddies ans also sharing in delicious. And certainly, thanks in your sweat!
dHtGZ5uCTLF
whoah this blog is great i love reading your articles. Keep up the good work! You know, many people are looking around for this information, you could aid them greatly.
I?d have to test with you here. Which isn’t one thing I often do! I take pleasure in reading a put up that will make folks think. Additionally, thanks for permitting me to remark!
This really answered my downside, thanks!
Hey there! Would you mind if I share your blog with my twitter group? There’s a lot of folks that I think would really enjoy your content. Please let me know. Thanks
hey there and thank you for your information ? I?ve definitely picked up something new from right here. I did however expertise some technical issues using this web site, as I experienced to reload the site many times previous to I could get it to load properly. I had been wondering if your hosting is OK? Not that I am complaining, but slow loading instances times will sometimes affect your placement in google and can damage your high-quality score if ads and marketing with Adwords. Anyway I?m adding this RSS to my e-mail and can look out for a lot more of your respective interesting content. Ensure that you update this again very soon..
I think other website proprietors should take this website as an model, very clean and magnificent user genial style and design, let alone the content. You are an expert in this topic!
We are a group of volunteers and opening a new scheme in our community. Your website offered us with valuable information to work on. You have done an impressive job and our entire community will be grateful to you.
Hi! Would you mind if I share your blog with my twitter group? There’s a lot of folks that I think would really enjoy your content. Please let me know. Thank you
Nice blog here! Also your web site loads up fast! What host are you using? Can I get your affiliate link to your host? I wish my website loaded up as quickly as yours lol
I do not even know how I finished up here, however I thought this post used to be good. I don’t recognize who you might be but certainly you are going to a well-known blogger in case you are not already 😉 Cheers!
Aw, this was a very nice post. In idea I wish to put in writing like this moreover ? taking time and actual effort to make a very good article? however what can I say? I procrastinate alot and certainly not seem to get one thing done.
I loved as much as you’ll receive carried out right here. The sketch is tasteful, your authored material stylish. nonetheless, you command get bought an edginess over that you wish be delivering the following. unwell unquestionably come further formerly again as exactly the same nearly a lot often inside case you shield this increase.
I have observed that online diploma is getting common because obtaining your degree online has become a popular solution for many people. Numerous people have definitely not had an opportunity to attend a regular college or university but seek the improved earning possibilities and a better job that a Bachelor Degree grants. Still other people might have a diploma in one field but want to pursue some thing they now have an interest in.
Hey, you used to write great, but the last few posts have been kinda boring? I miss your tremendous writings. Past several posts are just a little bit out of track! come on!
I’m really impressed with your writing skills as well as with the layout on your blog. Is this a paid theme or did you modify it yourself? Anyway keep up the excellent quality writing, it is rare to see a nice blog like this one these days..
Greetings! I’ve been following your site for a long time now and finally got the bravery to go ahead and give you a shout out from Kingwood Texas! Just wanted to tell you keep up the great work!
Hi there just wanted to give you a quick heads up and let you know a few of the pictures aren’t loading correctly. I’m not sure why but I think its a linking issue. I’ve tried it in two different web browsers and both show the same results.
Hello there, You’ve performed a fantastic job. I will definitely digg it and personally suggest to my friends. I am confident they’ll be benefited from this site.
Have you ever considered creating an e-book or guest authoring on other websites? I have a blog centered on the same ideas you discuss and would love to have you share some stories/information. I know my subscribers would enjoy your work. If you’re even remotely interested, feel free to send me an e-mail.
I’ve really noticed that repairing credit activity has to be conducted with techniques. If not, you might find yourself damaging your positioning. In order to be successful in fixing your credit ranking you have to take care that from this moment you pay your monthly dues promptly prior to their booked date. It is definitely significant on the grounds that by not accomplishing that area, all other actions that you will choose to adopt to improve your credit positioning will not be successful. Thanks for revealing your suggestions.
I have witnessed that rates for internet degree professionals tend to be a fantastic value. Like a full 4-year college Degree in Communication with the University of Phoenix Online consists of Sixty credits from $515/credit or $30,900. Also American Intercontinental University Online gives a Bachelors of Business Administration with a entire school feature of 180 units and a cost of $30,560. Online learning has made obtaining your college diploma so much easier because you may earn your degree in the comfort of your dwelling place and when you finish from office. Thanks for all the tips I have learned through the web site.
Hello there! This is my first visit to your blog! We are a team of volunteers and starting a new initiative in a community in the same niche. Your blog provided us beneficial information to work on. You have done a outstanding job!
I really appreciate this post. I?ve been looking everywhere for this! Thank goodness I found it on Bing. You have made my day! Thanks again
I just couldn’t depart your web site before suggesting that I extremely enjoyed the standard information a person provide for your visitors? Is gonna be back often in order to check up on new posts
Hello. excellent job. I did not imagine this. This is a fantastic story. Thanks!
Fantastic goods frdom you, man. I have take note your
stuff prior to annd you’resimply too magnificent.
I really like whazt you’ve boughtt riyht here, certainly
lke whaat yoou aare sayinng annd thee beet wway bby which yoou aree aying it.
You’re making it enjoyable and yyou stil take care of to tay iit smart.
I can’t wait too learn far more frtom you.
This iss really a terrific website.
You made some first rate points there. I seemed on the internet for the issue and found most individuals will go along with together with your website.
Good blog! I truly love how it is easy on my eyes and the data are well written. I am wondering how I might be notified when a new post has been made. I’ve subscribed to your feed which must do the trick! Have a nice day!
I was deeply moved by your post, which ignited a strong desire in me to explore the topic further. I have great admiration for your profound insights and expertise, and I genuinely appreciate your distinct perspective. Thank you for generously sharing your thoughts and dedicating your time to do so!Have a good one.
How’ve you been?We stumbled upon this page on a different website and thought we’d check it out. I’m liking what I see, so I’m going to follow you. Looking forward to further exploring your website.
I’m not sure why but this website is loading very slow for me. Is anyone else having this issue or is it a problem on my end? I’ll check back later and see if the problem still exists.
How’s it going?During our exploration of a new website, we came across a webpage that immediately caught our attention. We are extremely impressed with what we have seen so far and eagerly look forward to your upcoming updates. We are thrilled to continue exploring your website and uncovering all the amazing features it has to offer.Safe travels.
Hi there! This is kind of off topic but I need some advice from an established blog. Is it very difficult to set up your own blog? I’m not very techincal but I can figure things out pretty fast. I’m thinking about setting up my own but I’m not sure where to start. Do you have any points or suggestions? Many thanks
I haven?t checked in here for some time as I thought it was getting boring, but the last several posts are great quality so I guess I?ll add you back to my everyday bloglist. You deserve it my friend 🙂
I think other site proprietors should take this web site as an model, very clean and wonderful user genial style and design, let alone the content. You’re an expert in this topic!
Hiya! I just wish to give a huge thumbs up for the good information you’ve got here on this post. I will likely be coming again to your blog for extra soon.
What I have seen in terms of laptop or computer memory is the fact that there are specifications such as SDRAM, DDR and the like, that must fit in with the specs of the motherboard. If the computer’s motherboard is very current while there are no main system issues, modernizing the storage space literally will take under 1 hour. It’s among the list of easiest computer upgrade types of procedures one can picture. Thanks for giving your ideas.
Porn site
Viagra
Pornstar
Nice read!
Good job!
Thanks for your text. I would also love to say a health insurance broker also works well with the benefit of the actual coordinators of the group insurance plan. The health insurance professional is given a long list of benefits desired by someone or a group coordinator. What any broker does is try to find individuals or coordinators which usually best fit those desires. Then he shows his tips and if the two of you agree, the broker formulates binding agreement between the two parties.
Good ? I should definitely pronounce, impressed with your site. I had no trouble navigating through all the tabs as well as related information ended up being truly easy to do to access. I recently found what I hoped for before you know it at all. Quite unusual. Is likely to appreciate it for those who add forums or anything, website theme . a tones way for your customer to communicate. Excellent task..
One other issue is when you are in a circumstances where you would not have a co-signer then you may really need to try to exhaust all of your financial aid options. You can get many grants and other scholarship grants that will offer you finances to help with school expenses. Thanks a lot for the post.
You really make it appear really easy together with your presentation but I find this topic to be actually something that I feel I might by no means understand. It kind of feels too complicated and extremely wide for me. I am taking a look forward to your subsequent post, I will attempt to get the hang of it!
Admiring the dedication you put into your site and in depth information you offer. It’s awesome to come across a blog every once in a while that isn’t the same old rehashed information. Wonderful read! I’ve saved your site and I’m adding your RSS feeds to my Google account.
Hey very cool web site!! Man .. Excellent .. Amazing .. I’ll bookmark your website and take the feeds also?I’m happy to find numerous useful info here in the post, we need develop more techniques in this regard, thanks for sharing. . . . . .
Have you ever thought about writing an e-book or guest authoring on other blogs? I have a blog based upon on the same information you discuss and would really like to have you share some stories/information. I know my visitors would value your work. If you are even remotely interested, feel free to send me an e mail.
I have noticed that credit improvement activity must be conducted with tactics. If not, you will probably find yourself damaging your rating. In order to grow into success fixing your credit history you have to make sure that from this time you pay all of your monthly fees promptly before their timetabled date. It is definitely significant given that by never accomplishing so, all other activities that you will decide to try to improve your credit rank will not be useful. Thanks for revealing your thoughts.
Woah! I’m really loving the template/theme of this site. It’s simple, yet effective. A lot of times it’s difficult to get that “perfect balance” between usability and visual appeal. I must say you’ve done a fantastic job with this. Also, the blog loads extremely quick for me on Chrome. Excellent Blog!
An impressive share, I just given this onto a colleague who was doing slightly evaluation on this. And he in actual fact bought me breakfast as a result of I found it for him.. smile. So let me reword that: Thnx for the treat! But yeah Thnkx for spending the time to debate this, I feel strongly about it and love reading more on this topic. If doable, as you turn out to be expertise, would you mind updating your weblog with extra details? It’s highly helpful for me. Big thumb up for this blog post!
Holy cow! I’m in awe of the author’s writing skills and ability to convey complicated concepts in a concise and concise manner. This article is a true gem that deserves all the accolades it can get. Thank you so much, author, for providing your expertise and offering us with such a precious asset. I’m truly thankful!
Good write-up, I?m regular visitor of one?s blog, maintain up the excellent operate, and It is going to be a regular visitor for a long time.
Wow that was unusual. I just wrote an really long comment but after I clicked submit my comment didn’t show up. Grrrr… well I’m not writing all that over again. Anyhow, just wanted to say wonderful blog!
You actually make it seem so easy with your presentation however I find this matter to be actually one thing which I feel I might never understand. It seems too complicated and very broad for me. I am having a look ahead to your subsequent put up, I?ll attempt to get the grasp of it!
Thanks for this wonderful article. Also a thing is that the majority of digital cameras come equipped with some sort of zoom lens so that more or less of your scene to become included by simply ‘zooming’ in and out. These types of changes in {focus|focusing|concentration|target|the a**** length tend to be reflected inside the viewfinder and on significant display screen right on the back of the particular camera.
Thanks for the distinct tips shared on this blog. I have observed that many insurers offer clients generous reductions if they elect to insure a couple of cars together. A significant volume of households include several cars or trucks these days, specially those with more aged teenage young children still living at home, and the savings on policies can easily soon increase. So it makes sense to look for a bargain.
I was curious if you ever thought of changing the page layout of your blog? Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better. Youve got an awful lot of text for only having 1 or two pictures. Maybe you could space it out better?
It is appropriate time to make some plans for the future and it’s time to be happy. I’ve read this post and if I could I wish to suggest you few interesting things or suggestions. Perhaps you could write next articles referring to this article. I desire to read more things about it!
Hey very cool web site!! Man .. Beautiful .. Amazing .. I’ll bookmark your web site and take the feeds also?I am happy to find a lot of useful information here in the post, we need develop more techniques in this regard, thanks for sharing. . . . . .
I?m impressed, I must say. Really hardly ever do I encounter a blog that?s both educative and entertaining, and let me let you know, you have got hit the nail on the head. Your concept is outstanding; the difficulty is one thing that not sufficient people are speaking intelligently about. I am very happy that I stumbled throughout this in my seek for one thing relating to this.
JC0Ejmjb47I
Wow, this article is mind-blowing! The author has done a tremendous job of conveying the information in an engaging and enlightening manner. I can’t thank him enough for sharing such precious insights that have certainly enriched my understanding in this subject area. Kudos to him for producing such a masterpiece!
It?s arduous to search out educated folks on this topic, however you sound like you already know what you?re speaking about! Thanks
you are truly a just right webmaster. The website loading speed is incredible. It seems that you are doing any distinctive trick. Moreover, The contents are masterwork. you’ve done a magnificent job on this matter!
This really answered my downside, thank you!
With havin so much written content do you ever run into any problems of plagorism or copyright infringement? My website has a lot of exclusive content I’ve either written myself or outsourced but it looks like a lot of it is popping it up all over the internet without my agreement. Do you know any methods to help reduce content from being ripped off? I’d certainly appreciate it.
I think one of your advertisements triggered my browser to resize, you may well want to put that on your blacklist.
Hello there! This is my first visit to your blog! We are a group of volunteers and starting a new project in a community in the same niche. Your blog provided us valuable information to work on. You have done a extraordinary job!
Woah! I’m really loving the template/theme of this blog. It’s simple, yet effective. A lot of times it’s challenging to get that “perfect balance” between usability and appearance. I must say you have done a amazing job with this. Also, the blog loads extremely fast for me on Opera. Exceptional Blog!
Undeniably believe that which you stated. Your favorite reason appeared to be on the internet the simplest thing to be aware of. I say to you, I definitely get irked while people think about worries that they just do not know about. You managed to hit the nail upon the top and also defined out the whole thing without having side-effects , people could take a signal. Will likely be back to get more. Thanks
Please let me know if you’re looking for a article writer for your blog. You have some really good posts and I think I would be a good asset. If you ever want to take some of the load off, I’d absolutely love to write some articles for your blog in exchange for a link back to mine. Please shoot me an email if interested. Cheers!
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You clearly know what youre talking about, why throw away your intelligence on just posting videos to your blog when you could be giving us something informative to read?
It’s best to participate in a contest for probably the greatest blogs on the web. I’ll advocate this web site!
Wow that was strange. I just wrote an very long comment but after I clicked submit my comment didn’t appear. Grrrr… well I’m not writing all that over again. Anyhow, just wanted to say excellent blog!
I was more than happy to seek out this internet-site.I needed to thanks for your time for this glorious learn!! I positively having fun with every little bit of it and I’ve you bookmarked to check out new stuff you weblog post.
I will right away grab your rss feed as I can not find your e-mail subscription link or newsletter service. Do you’ve any? Kindly let me know so that I could subscribe. Thanks.
I just could not depart your site prior to suggesting that I really enjoyed the standard info a person provide for your visitors? Is gonna be back often to check up on new posts
Do you mind if I quote a few of your posts as long as I provide credit and sources back to your blog? My blog is in the very same area of interest as yours and my users would really benefit from a lot of the information you provide here. Please let me know if this ok with you. Appreciate it!
Hiya! I simply would like to give an enormous thumbs up for the good information you’ve got right here on this post. I will probably be coming back to your blog for more soon.
Try Bht club now
I do not even know the way I finished up right here, however I thought this submit used to be great. I do not recognize who you’re but certainly you’re going to a famous blogger if you are not already 😉 Cheers!
colour prediction app
Many thanks for your post. I’d really like to comment that the price of car insurance will vary from one policy to another, mainly because there are so many different issues which give rise to the overall cost. For instance, the make and model of the car or truck will have a huge bearing on the charge. A reliable aged family car will have a more affordable premium over a flashy sports vehicle.
66 lottery is a colour trading platform
What?s Happening i am new to this, I stumbled upon this I have found It absolutely helpful and it has helped me out loads. I hope to contribute & aid other users like its aided me. Great job.
I know this if off topic but I’m looking into starting my own weblog and was curious what all is needed to get set up? I’m assuming having a blog like yours would cost a pretty penny? I’m not very web savvy so I’m not 100 positive. Any recommendations or advice would be greatly appreciated. Cheers
I will right away grab your rss as I can not find your email subscription link or newsletter service. Do you have any? Please let me know in order that I could subscribe. Thanks.
colour prediction app
login on 66 lottery and start your journey
66 lottery is a colour trading platform
Thanks for your tips on this blog. A single thing I would want to say is always that purchasing electronics items over the Internet is not new. In reality, in the past decades alone, the market for online gadgets has grown considerably. Today, you will discover practically almost any electronic tool and other gadgets on the Internet, including cameras as well as camcorders to computer pieces and gambling consoles.
Thank you for the sensible critique. Me and my neighbor were just preparing to do some research about this. We got a grab a book from our local library but I think I learned more from this post. I’m very glad to see such fantastic info being shared freely out there.
I have observed that in the world these days, video games are classified as the latest fad with kids of all ages. There are times when it may be out of the question to drag young kids away from the games. If you want the best of both worlds, there are several educational video games for kids. Interesting post.
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You definitely know what youre talking about, why waste your intelligence on just posting videos to your weblog when you could be giving us something informative to read?
Try 66lottery now
I’m not sure why but this weblog is loading very slow for me. Is anyone else having this problem or is it a problem on my end? I’ll check back later on and see if the problem still exists.
colour prediction app
Excellent beat ! I wish to apprentice while you amend your site, how can i subscribe for a blog website? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast provided bright clear concept
Try jalwa game now
Try jalwa game now
It?s actually a cool and useful piece of information. I am satisfied that you simply shared this useful info with us. Please stay us informed like this. Thank you for sharing.
It?s actually a great and helpful piece of information. I?m glad that you simply shared this useful information with us. Please stay us up to date like this. Thanks for sharing.
I was suggested this website by my cousin. I’m not sure whether this post is written by him as nobody else know such detailed about my problem. You’re wonderful! Thanks!
We are a gaggle of volunteers and starting a new scheme in our community. Your site offered us with helpful information to paintings on. You have done an impressive process and our entire neighborhood will probably be thankful to you.
My brother suggested I might like this web site. He was entirely right. This post actually made my day. You can not imagine just how much time I had spent for this info! Thanks!
Hi there! This is kind of off topic but I need some guidance from an established blog. Is it very difficult to set up your own blog? I’m not very techincal but I can figure things out pretty quick. I’m thinking about creating my own but I’m not sure where to start. Do you have any ideas or suggestions? Thanks
I am not sure where you are getting your info, but great topic. I needs to spend some time learning much more or understanding more. Thanks for wonderful info I was looking for this information for my mission.
This blog doesn’t display correctly on my i phone – you might wanna try and fix that
Thanks for the guidelines you have discussed here. One more thing I would like to express is that laptop or computer memory requirements generally go up along with other developments in the engineering. For instance, whenever new generations of cpus are brought to the market, there is usually an equivalent increase in the dimensions demands of both the personal computer memory and also hard drive room. This is because the software program operated by means of these processor chips will inevitably increase in power to take advantage of the new technological know-how.
hentai porn download
Thanks for your beneficial post. In recent times, I have been able to understand that the symptoms of mesothelioma cancer are caused by the actual build up of fluid between lining in the lung and the chest cavity. The sickness may start while in the chest spot and get distributed to other areas of the body. Other symptoms of pleural mesothelioma cancer include weight-loss, severe respiration trouble, fever, difficulty taking in food, and puffiness of the face and neck areas. It must be noted that some people with the disease usually do not experience virtually any serious signs at all.
downoad sex games
Thank you, I’ve recently been looking for info about this subject matter for ages and yours is the best I have located so far.
I would like to thank you for the efforts you have put in writing this web site. I’m hoping the same high-grade website post from you in the upcoming as well. Actually your creative writing skills has inspired me to get my own site now. Actually the blogging is spreading its wings rapidly. Your write up is a great example of it.
I’ve been browsing online more than 3 hours today, yet I never found any interesting article like yours. It?s pretty worth enough for me. Personally, if all web owners and bloggers made good content as you did, the net will be much more useful than ever before.
Do you have a spam issue on this blog; I also am a blogger, and I was wanting to know your situation; we have created some nice procedures and we are looking to exchange strategies with other folks, why not shoot me an e-mail if interested.
I discovered more new stuff on this weight-loss issue. 1 issue is that good nutrition is vital if dieting. A big reduction in bad foods, sugary foodstuff, fried foods, sweet foods, red meat, and bright flour products can be necessary. Holding wastes parasites, and contaminants may prevent aims for fat loss. While selected drugs momentarily solve the situation, the unpleasant side effects are not worth it, and so they never provide more than a short-term solution. This can be a known proven fact that 95 of dietary fads fail. Many thanks sharing your opinions on this site.
After examine just a few of the weblog posts on your website now, and I truly like your method of blogging. I bookmarked it to my bookmark website record and shall be checking again soon. Pls check out my site as well and let me know what you think.
I will right away grab your rss feed as I can not find your e-mail subscription link or e-newsletter service. Do you’ve any? Kindly let me know so that I could subscribe. Thanks.
Thanks for another magnificent post. Where else may just anybody get that type of info in such a perfect manner of writing? I’ve a presentation subsequent week, and I’m at the look for such info.
A person essentially assist to make seriously articles I’d state. This is the very first time I frequented your website page and thus far? I surprised with the research you made to make this actual put up amazing. Great activity!
You made some first rate factors there. I regarded on the internet for the difficulty and found most people will associate with along with your website.
I would like to thank you for the efforts you’ve put in writing this web site. I am hoping the same high-grade blog post from you in the upcoming as well. In fact your creative writing abilities has encouraged me to get my own web site now. Actually the blogging is spreading its wings rapidly. Your write up is a great example of it.
Thank you for the good writeup. It actually used to be a entertainment account it. Look advanced to more brought agreeable from you! By the way, how could we keep in touch?
Wow, superb weblog layout! How lengthy have you ever been running a blog for? you make blogging look easy. The full glance of your site is great, as neatly as the content!
Hmm it looks like your website ate my first comment (it was extremely long) so I guess I’ll just sum it up what I submitted and say, I’m thoroughly enjoying your blog. I too am an aspiring blog blogger but I’m still new to the whole thing. Do you have any recommendations for inexperienced blog writers? I’d genuinely appreciate it.
Thanks for the tips you discuss through this website. In addition, numerous young women which become pregnant will not even seek to get health care insurance because they are full of fearfulness they might not qualify. Although some states now require that insurers produce coverage in spite of the pre-existing conditions. Charges on these kind of guaranteed plans are usually larger, but when considering the high cost of health care it may be a new safer approach to take to protect your financial future.
F*ckin? amazing things here. I am very glad to look your post. Thanks a lot and i am taking a look ahead to contact you. Will you please drop me a e-mail?
you’re in point of fact a just right webmaster. The web site loading pace is amazing. It sort of feels that you’re doing any distinctive trick. In addition, The contents are masterwork. you have done a great task in this matter!
you are really a good webmaster. The web site loading velocity is incredible. It kind of feels that you are doing any unique trick. In addition, The contents are masterwork. you have done a great activity on this topic!
Yet another thing I would like to convey is that as opposed to trying to fit all your online degree classes on days of the week that you finish work (because most people are fatigued when they come home), try to get most of your instructional classes on the week-ends and only a couple courses in weekdays, even if it means a little time away from your saturdays. This is really good because on the saturdays and sundays, you will be far more rested and concentrated on school work. Thanks a bunch for the different points I have learned from your weblog.
Howdy are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and set up my own. Do you need any coding knowledge to make your own blog? Any help would be really appreciated!
You must take part in a contest for probably the greatest blogs on the web. I will recommend this site!
My spouse and I absolutely love your blog and find most of your post’s to be just what I’m looking for. Does one offer guest writers to write content for you? I wouldn’t mind writing a post or elaborating on some of the subjects you write about here. Again, awesome web site!
Today, with the fast way of living that everyone leads, credit cards get this amazing demand throughout the economy. Persons coming from every area are using credit card and people who not using the credit cards have prepared to apply for just one. Thanks for spreading your ideas about credit cards.
My partner and I absolutely love your blog and find nearly all of your post’s to be exactly what I’m looking for. Do you offer guest writers to write content for yourself? I wouldn’t mind publishing a post or elaborating on a number of the subjects you write about here. Again, awesome web site!
Hey just wanted to give you a quick heads up and let you know a few of the pictures aren’t loading properly. I’m not sure why but I think its a linking issue. I’ve tried it in two different web browsers and both show the same outcome.
Wow, wonderful weblog format! How lengthy have you ever been blogging for? you make blogging glance easy. The entire look of your website is fantastic, as well as the content material!
Pornstar
A person essentially help to make seriously articles I would state. This is the very first time I frequented your website page and thus far? I surprised with the research you made to make this particular publish incredible. Magnificent job!
Buy Drugs
One thing I’d really like to say is that before obtaining more personal computer memory, take a look at the machine in which it will be installed. In case the machine is running Windows XP, for instance, the actual memory limit is 3.25GB. Using above this would merely constitute just a waste. Be sure that one’s mother board can handle your upgrade amount, as well. Great blog post.
I haven?t checked in here for some time since I thought it was getting boring, but the last few posts are good quality so I guess I?ll add you back to my everyday bloglist. You deserve it my friend 🙂
I loved as much as you will receive carried out right here. The sketch is attractive, your authored material stylish. nonetheless, you command get bought an shakiness over that you wish be delivering the following. unwell unquestionably come more formerly again as exactly the same nearly a lot often inside case you shield this increase.
Hmm it seems like your website ate my first comment (it was extremely long) so I guess I’ll just sum it up what I submitted and say, I’m thoroughly enjoying your blog. I too am an aspiring blog writer but I’m still new to everything. Do you have any points for newbie blog writers? I’d really appreciate it.
Scam
Sex
I have acquired some new elements from your site about pcs. Another thing I’ve always thought is that computer systems have become a specific thing that each family must have for several reasons. They offer convenient ways in which to organize the home, pay bills, search for information, study, hear music and in many cases watch television shows. An innovative technique to complete all of these tasks is to use a mobile computer. These computers are mobile, small, potent and mobile.
Pornstar
Buy Drugs
I like the valuable information you supply to your articles. I will bookmark your blog and check again right here regularly. I’m fairly certain I will learn many new stuff proper right here! Good luck for the following!
Porn
With havin so much written content do you ever run into any problems of plagorism or copyright infringement? My blog has a lot of completely unique content I’ve either created myself or outsourced but it appears a lot of it is popping it up all over the web without my authorization. Do you know any methods to help prevent content from being ripped off? I’d certainly appreciate it.
Scam
Viagra
Pornstar
{
Thinking about releasing equity from your home? Review top lenders and understand your rights and obligations before making a decision.
Porn
Porn
Buy Drugs
Buy Drugs
Uncertain whether a secured loan is right for you? Explore the benefits, such as lower interest rates and larger borrowing amounts.
Release the value in your property with a reliable home equity loan — ideal for covering home improvements, large expenses, or refinancing.
Scam
Equity release solutions may provide the financial freedom you’ve been looking for. Learn how to use the equity tied up in your home without having to downsize.
Thinking about releasing equity from your home? Review top lenders and learn about your rights and obligations before making a decision.
Hello are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and set up my own. Do you need any html coding knowledge to make your own blog? Any help would be greatly appreciated!
Considering releasing equity from your home? Review top lenders and learn about your financial responsibilities before making a decision.
Hey there! This is my first comment here so I just wanted to give a quick shout out and tell you I truly enjoy reading your posts. Can you suggest any other blogs/websites/forums that cover the same subjects? Many thanks!
Equity release solutions may provide the financial freedom you’ve been looking for. Learn how to tap into the equity tied up in your home without having to move.
Thinking about releasing equity from your home? Review top lenders and learn about your rights and obligations before making a decision.
Buy Drugs
Release the value in your property with a secure home equity loan — ideal for covering home improvements, major purchases, or debt consolidation.
Porn
Viagra
Equity release solutions may provide the financial freedom you’ve been looking for. Learn how to use the equity tied up in your home without having to move.
Thinking about releasing equity from your home? Review top lenders and learn about your rights and obligations before making a decision.
I have really learned newer and more effective things by your website. One other thing I would like to say is always that newer personal computer os’s are inclined to allow additional memory for use, but they likewise demand more ram simply to function. If an individual’s computer can’t handle extra memory and also the newest computer software requires that memory increase, it could be the time to shop for a new Laptop or computer. Thanks
Are you considering a loan against your home to manage your financial obligations? Find out more and check what options may be available to you.
Equity release solutions may provide the financial support you’ve been needing. Learn how to tap into the equity tied up in your home without having to downsize.
You should take part in a contest for one of the best sites on the net. I’m going to highly recommend this blog!
Howdy would you mind letting me know which web host you’re using? I’ve loaded your blog in 3 completely different web browsers and I must say this blog loads a lot quicker then most. Can you recommend a good web hosting provider at a fair price? Cheers, I appreciate it!
Discover how a homeowner loan can help you access the money you need without selling your home. Compare lenders and tailor a plan that fits your needs.
Buy Drugs
Release the equity in your property with a secure home equity loan — ideal for covering home improvements, major purchases, or refinancing.
Discover how a secured loan can help you access the money you need without parting with your home. Review lenders and tailor a plan that fits your needs.
Home equity release may provide the financial freedom you’ve been looking for. Learn how to use the equity tied up in your home without having to move.
Home equity release may provide the financial support you’ve been looking for. Learn how to tap into the equity tied up in your home without having to move.
Considering releasing equity from your home? Review top lenders and understand your rights and obligations before making a decision.
It is in reality a great and helpful piece of info. I am satisfied that you shared this helpful info with us. Please keep us informed like this. Thank you for sharing.
Porn site
Hello! I simply would like to give a huge thumbs up for the nice data you might have here on this post. I will likely be coming back to your weblog for extra soon.
Sex
Excellent post however , I was wanting to know if you could write a litte more on this topic? I’d be very grateful if you could elaborate a little bit more. Appreciate it!
Discover how a homeowner loan can help you access the money you need without parting with your home. Compare lenders and customise a plan that fits your needs.
I appreciate your wp design, wherever do you obtain it through?
It?s in point of fact a great and useful piece of info. I am glad that you simply shared this useful information with us. Please keep us informed like this. Thank you for sharing.
If you’re a homeowner looking to borrow money, a secured loan could be a sensible option. Access better rates by using your home as collateral.
You may be able to secure larger loans and enjoy lower monthly repayments by taking out a loan secured on your property. Review current offers today.
Uncertain whether a secured loan is right for you? Explore the benefits, such as more favourable terms and larger borrowing amounts.
You may be able to borrow more and enjoy better interest rates by using your home’s equity. Review current offers today.
Everything is very open with a really clear explanation of the challenges. It was truly informative. Your site is very helpful. Thank you for sharing!
If you’re a property owner looking to get a loan, a secured loan could be a wise option. Leverage better rates by using your home as collateral.
Equity release solutions may provide the financial support you’ve been looking for. Learn how to use the equity tied up in your home without having to downsize.
I?m not sure where you’re getting your info, but good topic. I needs to spend some time learning much more or understanding more. Thanks for fantastic information I was looking for this info for my mission.
In this great scheme of things you’ll receive a B- with regard to hard work. Exactly where you actually misplaced us was on the details. As it is said, the devil is in the details… And it could not be much more correct at this point. Having said that, let me inform you just what did deliver the results. The writing is definitely incredibly convincing and this is probably why I am taking the effort in order to comment. I do not make it a regular habit of doing that. Next, whilst I can easily see the jumps in reasoning you make, I am not certain of just how you seem to unite the ideas which make the final result. For now I shall subscribe to your issue but hope in the future you connect the facts better.
Excellent blog right here! Additionally your web site loads up very fast! What web host are you the use of? Can I get your associate link in your host? I wish my site loaded up as fast as yours lol
After checking out a number of the blog posts on your web page, I really like your way of blogging. I bookmarked it to my bookmark website list and will be checking back soon. Take a look at my website too and tell me your opinion.
It’s a pity you don’t have a donate button! I’d certainly donate to this excellent blog! I guess for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to fresh updates and will talk about this website with my Facebook group. Chat soon!
Porn
I have viewed that clever real estate agents almost everywhere are Advertising and marketing. They are realizing that it’s in addition to placing a poster in the front area. It’s really in relation to building connections with these sellers who at some time will become buyers. So, once you give your time and efforts to assisting these vendors go it alone — the “Law involving Reciprocity” kicks in. Great blog post.
Buy Drugs
I’ve been surfing online greater than 3 hours these days, but I by no means discovered any fascinating article like yours. It is beautiful worth sufficient for me. In my view, if all site owners and bloggers made just right content material as you probably did, the web will likely be much more helpful than ever before.
Thanks for sharing superb informations. Your web-site is very cool. I’m impressed by the details that you?ve on this web site. It reveals how nicely you understand this subject. Bookmarked this web page, will come back for extra articles. You, my friend, ROCK! I found just the information I already searched everywhere and simply couldn’t come across. What an ideal web-site.
What i don’t realize is actually how you’re not actually much more well-liked than you might be right now. You’re so intelligent. You realize therefore considerably relating to this subject, made me personally consider it from numerous varied angles. Its like women and men aren’t fascinated unless it is one thing to accomplish with Lady gaga! Your own stuffs great. Always maintain it up!
Sex
Valuable info. Lucky me I found your web site by accident, and I’m shocked why this accident didn’t happened earlier! I bookmarked it.
you’re really a good webmaster. The web site loading speed is amazing. It seems that you are doing any unique trick. Moreover, The contents are masterpiece. you’ve done a magnificent job on this topic!
I cherished as much as you will receive carried out right here. The sketch is attractive, your authored subject matter stylish. however, you command get got an nervousness over that you want be handing over the following. sick without a doubt come more before again as exactly the similar just about very frequently inside case you defend this increase.
Thanks , I’ve recently been looking for information about this subject for ages and yours is the best I’ve discovered till now. But, what about the conclusion? Are you sure about the source?
Thanks for another excellent article. Where else could anyone get that type of information in such a perfect way of writing? I’ve a presentation next week, and I’m on the look for such information.
Another important aspect is that if you are an older person, travel insurance for pensioners is something you ought to really look at. The more mature you are, the more at risk you are for having something negative happen to you while in another country. If you are never covered by quite a few comprehensive insurance, you could have a number of serious troubles. Thanks for discussing your advice on this blog site.
What i don’t realize is in reality how you are no longer actually much more smartly-preferred than you might be now. You are so intelligent. You recognize thus significantly in terms of this matter, produced me personally consider it from numerous varied angles. Its like men and women don’t seem to be fascinated until it?s something to accomplish with Woman gaga! Your personal stuffs excellent. Always handle it up!
I think this is one of the most vital info for me. And i am glad reading your article. But wanna remark on few general things, The web site style is perfect, the articles is really nice : D. Good job, cheers
Viagra
Hey there, I think your blog might be having browser compatibility issues. When I look at your blog site in Safari, it looks fine but when opening in Internet Explorer, it has some overlapping. I just wanted to give you a quick heads up! Other then that, excellent blog!
Thanks a bunch for sharing this with all folks you actually realize what you’re speaking about! Bookmarked. Please also discuss with my site =). We could have a link alternate contract between us!
Porn
An added important aspect is that if you are an older person, travel insurance regarding pensioners is something you ought to really contemplate. The older you are, the greater at risk you are for allowing something undesirable happen to you while in foreign countries. If you are not necessarily covered by quite a few comprehensive insurance plan, you could have quite a few serious problems. Thanks for sharing your hints on this web site.
Good day! This is my first visit to your blog! We are a collection of volunteers and starting a new project in a community in the same niche. Your blog provided us useful information to work on. You have done a extraordinary job!
Based on my observation, after a property foreclosure home is marketed at a sale, it is common with the borrower to be able to still have some sort ofthat remaining balance on the personal loan. There are many financial institutions who attempt to have all fees and liens cleared by the subsequent buyer. Even so, depending on a number of programs, legislation, and state legal guidelines there may be several loans that are not easily sorted out through the exchange of loans. Therefore, the duty still rests on the lender that has acquired his or her property in foreclosure process. Many thanks sharing your ideas on this blog.
I have been browsing online greater than three hours lately, but I never discovered any fascinating article like yours. It is lovely price sufficient for me. In my view, if all website owners and bloggers made just right content material as you probably did, the internet might be much more helpful than ever before.
It’s the best time to make some plans for the future and it’s time to be happy. I’ve read this post and if I could I wish to suggest you few interesting things or suggestions. Perhaps you can write next articles referring to this article. I wish to read even more things about it!
Thanks for revealing your ideas with this blog. Furthermore, a fantasy regarding the banks intentions any time talking about foreclosures is that the bank will not getreceive my payments. There is a fair bit of time which the bank will take payments occasionally. If you are as well deep inside the hole, they are going to commonly demand that you pay the payment fully. However, that doesn’t mean that they will have any sort of payments at all. If you and the loan company can seem to work something out, your foreclosure procedure may cease. However, if you ever continue to pass up payments within the new plan, the property foreclosure process can just pick up from where it left off.
Scam
Thanks for the concepts you have discussed here. Furthermore, I believe there are several factors which will keep your car insurance policy premium down. One is, to consider buying cars and trucks that are from the good report on car insurance providers. Cars which have been expensive are usually more at risk of being stolen. Aside from that insurance coverage is also good value of the car, so the more pricey it is, then higher the premium you pay.
Thank you, I’ve recently been looking for info about this subject for ages and yours is the greatest I have discovered so far. But, what about the conclusion? Are you sure about the source?
Generally I don’t read post on blogs, but I wish to say that this write-up very forced me to try and do it! Your writing style has been amazed me. Thanks, quite nice post.
Have you ever considered creating an e-book or guest authoring on other blogs? I have a blog centered on the same topics you discuss and would really like to have you share some stories/information. I know my audience would enjoy your work. If you are even remotely interested, feel free to shoot me an email.
whoah this blog is magnificent i love reading your articles. Keep up the good work! You know, a lot of people are looking around for this info, you can help them greatly.
Thanks for enabling me to attain new suggestions about computer systems. I also possess the belief that certain of the best ways to keep your laptop in excellent condition has been a hard plastic-type material case, or maybe shell, which fits over the top of the computer. These types of protective gear tend to be model targeted since they are manufactured to fit perfectly across the natural covering. You can buy all of them directly from owner, or through third party places if they are designed for your laptop, however only a few laptop will have a shell on the market. All over again, thanks for your recommendations.
Hey there! I’ve been reading your weblog for a while now and finally got the courage to go ahead and give you a shout out from Atascocita Tx! Just wanted to tell you keep up the excellent work!
I think this is among the most vital information for me. And i’m glad reading your article. But should remark on few general things, The website style is perfect, the articles is really excellent : D. Good job, cheers
Thanks for the new things you have revealed in your article. One thing I would really like to discuss is that FSBO interactions are built over time. By introducing yourself to the owners the first saturday and sunday their FSBO will be announced, prior to masses begin calling on Monday, you create a good interconnection. By mailing them tools, educational products, free reviews, and forms, you become a good ally. By taking a personal affinity for them along with their problem, you create a solid link that, on most occasions, pays off once the owners decide to go with a realtor they know and trust – preferably you actually.
Hello! I just wish to give a huge thumbs up for the great information you have right here on this post. I might be coming back to your weblog for more soon.
Good day! This is kind of off topic but I need some guidance from an established blog. Is it tough to set up your own blog? I’m not very techincal but I can figure things out pretty quick. I’m thinking about creating my own but I’m not sure where to begin. Do you have any ideas or suggestions? Many thanks
great issues altogether, you simply received a new reader. What would you suggest in regards to your post that you made some days in the past? Any certain?
Just wish to say your article is as surprising. The clarity in your post is simply excellent and i can assume you’re an expert on this subject. Fine with your permission let me to grab your RSS feed to keep up to date with forthcoming post. Thanks a million and please continue the enjoyable work.
Undeniably believe that which you said. Your favorite justification appeared to be on the net the easiest thing to be aware of. I say to you, I certainly get irked while people consider worries that they just do not know about. You managed to hit the nail upon the top and also defined out the whole thing without having side effect , people can take a signal. Will probably be back to get more. Thanks
In line with my observation, after a foreclosed home is offered at an auction, it is common with the borrower in order to still have any remaining balance on the mortgage. There are many financial institutions who aim to have all costs and liens paid off by the subsequent buyer. Nevertheless, depending on particular programs, legislation, and state regulations there may be a few loans which aren’t easily resolved through the shift of lending options. Therefore, the obligation still lies on the borrower that has had his or her property in foreclosure process. Thank you for sharing your notions on this site.
Thanks for expressing your ideas. I’d personally also like to express that video games have been actually evolving. Today’s technology and inventions have aided create realistic and active games. These kinds of entertainment video games were not as sensible when the actual concept was first of all being used. Just like other areas of electronics, video games also have had to grow by way of many decades. This is testimony for the fast growth of video games.
I’m truly enjoying the design and layout of your blog. It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often. Did you hire out a developer to create your theme? Excellent work!
Hey there! I know this is kinda off topic but I was wondering if you knew where I could get a captcha plugin for my comment form? I’m using the same blog platform as yours and I’m having problems finding one? Thanks a lot!
Thank you for the good writeup. It in fact was a amusement account it. Look advanced to more added agreeable from you! By the way, how can we communicate?
Hey there would you mind letting me know which hosting company you’re utilizing? I’ve loaded your blog in 3 completely different web browsers and I must say this blog loads a lot quicker then most. Can you suggest a good internet hosting provider at a honest price? Thanks, I appreciate it!
I would also like to add that in case you do not currently have an insurance policy or else you do not belong to any group insurance, chances are you’ll well really benefit from seeking aid from a health insurance broker. Self-employed or those that have medical conditions typically seek the help of the health insurance brokerage. Thanks for your writing.
obviously like your website however you need to test the spelling on several of your posts. Many of them are rife with spelling problems and I find it very troublesome to inform the truth then again I will certainly come back again.
I know this if off topic but I’m looking into starting my own weblog and was curious what all is required to get set up? I’m assuming having a blog like yours would cost a pretty penny? I’m not very internet savvy so I’m not 100 sure. Any recommendations or advice would be greatly appreciated. Appreciate it
Superb post but I was wondering if you could write a litte more on this subject? I’d be very thankful if you could elaborate a little bit more. Many thanks!
I do accept as true with all of the ideas you’ve presented to your post. They are really convincing and can certainly work. Nonetheless, the posts are very brief for beginners. May just you please prolong them a little from next time? Thank you for the post.
Interesting article. It’s very unfortunate that over the last decade, the travel industry has already been able to to handle terrorism, SARS, tsunamis, bird flu, swine flu, as well as first ever true global economic depression. Through all of it the industry has proven to be strong, resilient as well as dynamic, obtaining new tips on how to deal with trouble. There are often fresh troubles and opportunities to which the field must yet again adapt and respond.
I’m not sure exactly why but this website is loading incredibly slow for me. Is anyone else having this problem or is it a problem on my end? I’ll check back later on and see if the problem still exists.
Hi are using WordPress for your site platform? I’m new to the blog world but I’m trying to get started and set up my own. Do you need any html coding knowledge to make your own blog? Any help would be really appreciated!
Nice post. I used to be checking constantly this weblog and I am inspired! Very useful info specifically the closing phase 🙂 I care for such information much. I used to be looking for this particular info for a very long time. Thanks and best of luck.
Fantastic beat ! I would like to apprentice while you amend your site, how could i subscribe for a blog website? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast provided bright clear idea
Heya i?m for the primary time here. I found this board and I find It really useful & it helped me out much. I’m hoping to give one thing back and aid others such as you helped me.
you’re really a good webmaster. The site loading speed is amazing. It seems that you’re doing any unique trick. In addition, The contents are masterpiece. you have done a excellent job on this topic!
One thing is the fact that one of the most widespread incentives for utilizing your card is a cash-back or perhaps rebate offer. Generally, you’ll get 1-5 back for various acquisitions. Depending on the cards, you may get 1 in return on most buying, and 5 in return on expenditures made at convenience stores, gasoline stations, grocery stores in addition to ‘member merchants’.
Its like you read my mind! You appear to know so much about this, like you wrote the book in it or something. I think that you can do with some pics to drive the message home a bit, but instead of that, this is fantastic blog. A great read. I’ll definitely be back.
whoah this blog is excellent i love studying your posts. Keep up the great paintings! You realize, lots of persons are looking around for this information, you can help them greatly.
I do not even understand how I ended up right here, but I believed this publish was once great. I do not know who you are however certainly you’re going to a famous blogger when you aren’t already 😉 Cheers!
I believe that avoiding processed foods would be the first step in order to lose weight. They may taste fine, but prepared foods include very little nutritional value, making you try to eat more just to have enough energy to get with the day. For anyone who is constantly consuming these foods, moving over to cereals and other complex carbohydrates will help you have more energy while eating less. Interesting blog post.
Wow that was unusual. I just wrote an very long comment but after I clicked submit my comment didn’t appear. Grrrr… well I’m not writing all that over again. Anyhow, just wanted to say wonderful blog!
Thanks a bunch for sharing this with all people you really recognise what you are speaking approximately! Bookmarked. Kindly also seek advice from my website =). We can have a link change contract between us!
I can’t express how much I admire the effort the author has put into creating this exceptional piece of content. The clarity of the writing, the depth of analysis, and the abundance of information presented are simply astonishing. Her passion for the subject is obvious, and it has certainly resonated with me. Thank you, author, for providing your wisdom and enriching our lives with this extraordinary article!
great points altogether, you just gained a brand new reader. What would you recommend in regards to your post that you made a few days ago? Any positive?
I think this is one of the most significant info for me. And i’m glad reading your article. But should remark on some general things, The site style is ideal, the articles is really great : D. Good job, cheers
Excellent website. A lot of useful information here. I?m sending it to some pals ans additionally sharing in delicious. And obviously, thank you in your sweat!
Thanks for your write-up. Another item is that just being a photographer consists of not only difficulties in taking award-winning photographs but additionally hardships in establishing the best digital camera suited to your requirements and most especially hardships in maintaining the grade of your camera. It is very correct and apparent for those professional photographers that are in capturing the nature’s exciting scenes — the mountains, the particular forests, the particular wild and the seas. Going to these amazing places undoubtedly requires a digital camera that can meet the wild’s hard areas.
Does your blog have a contact page? I’m having a tough time locating it but, I’d like to send you an email. I’ve got some ideas for your blog you might be interested in hearing. Either way, great blog and I look forward to seeing it develop over time.
Thanks for the tips you have discussed here. Moreover, I believe there are many factors that really keep your car insurance premium decrease. One is, to think about buying motors that are inside good directory of car insurance organizations. Cars that are expensive will be more at risk of being robbed. Aside from that insurance is also in accordance with the value of the car, so the costlier it is, then the higher a premium you make payment for.
certainly like your web-site but you have to check the spelling on quite a few of your posts. Many of them are rife with spelling problems and I find it very troublesome to tell the truth nevertheless I will surely come back again.
Spot on with this write-up, I really think this amazing site needs far more attention. I’ll probably be back again to see more, thanks for the advice!
You ought to be a part of a contest for one of the most useful sites on the web. I’m going to highly recommend this site!
I’m amazed, I must say. Rarely do I come across a blog that’s equally educative and interesting, and let me tell you, you’ve hit the nail on the head. The problem is an issue that not enough folks are speaking intelligently about. I’m very happy I came across this in my search for something concerning this.
I used to be able to find good info from your content.
Very good article. I will be facing a few of these issues as well..
Aw, this was a very nice post. Taking the time and actual effort to make a very good article… but what can I say… I hesitate a lot and never seem to get anything done.
Very good info. Lucky me I recently found your website by chance (stumbleupon). I’ve book-marked it for later!
A fascinating discussion is worth comment. I think that you should publish more on this subject, it might not be a taboo subject but generally people don’t speak about such issues. To the next! Kind regards!
This is a topic that’s close to my heart… Many thanks! Exactly where can I find the contact details for questions?
Terrific paintings! That is the type of info that should be shared around the net. Shame on Google for now not positioning this post upper! Come on over and talk over with my site . Thank you =)
hey there and thanks on your information ? I?ve certainly picked up something new from proper here. I did however expertise some technical issues using this website, since I skilled to reload the website many occasions previous to I could get it to load properly. I had been wondering in case your web hosting is OK? No longer that I am complaining, but slow loading circumstances instances will very frequently impact your placement in google and can harm your quality ranking if ads and ***********|advertising|advertising|advertising and *********** with Adwords. Well I am including this RSS to my e-mail and can look out for much more of your respective interesting content. Ensure that you update this again very soon..
I was able to find good info from your blog posts.
One more important aspect is that if you are a mature person, travel insurance pertaining to pensioners is something you ought to really think about. The elderly you are, the harder at risk you happen to be for allowing something undesirable happen to you while abroad. If you are not covered by several comprehensive insurance policy, you could have quite a few serious issues. Thanks for sharing your guidelines on this web site.
One thing I’d like to comment on is that fat reduction plan fast may be possible by the proper diet and exercise. Ones size not only affects appearance, but also the quality of life. Self-esteem, depression, health risks, and also physical ability are damaged in weight gain. It is possible to make everything right and at the same time having a gain. In such a circumstance, a problem may be the primary cause. While a lot food and never enough physical exercise are usually to blame, common medical conditions and trusted prescriptions may greatly add to size. Thx for your post here.
This web page is mostly a stroll-by for the entire data you wanted about this and didn?t know who to ask. Glimpse here, and you?ll definitely discover it.
I?m impressed, I need to say. Actually rarely do I encounter a weblog that?s each educative and entertaining, and let me inform you, you may have hit the nail on the head. Your concept is excellent; the issue is one thing that not enough people are talking intelligently about. I’m very glad that I stumbled throughout this in my seek for one thing regarding this.
F*ckin? remarkable issues here. I?m very glad to see your article. Thanks so much and i am looking ahead to touch you. Will you kindly drop me a e-mail?
Such a practical bit of content.
This article is valuable.
This is the kind of post I truly appreciate.
I gained useful knowledge from this.
I’ll surely return to read more.
The clarity in this piece is praiseworthy.
Heya this is kinda of off topic but I was wondering if blogs use WYSIWYG editors or if you have to manually code with HTML. I’m starting a blog soon but have no coding skills so I wanted to get advice from someone with experience. Any help would be greatly appreciated!
I truly enjoyed the way this was written.
Aw, this was a very nice post. In concept I want to put in writing like this moreover ? taking time and precise effort to make an excellent article? but what can I say? I procrastinate alot and under no circumstances appear to get something done.
I just could not leave your web site prior to suggesting that I really enjoyed the standard information an individual supply in your guests? Is going to be again incessantly to check out new posts
Thank you for the auspicious writeup. It in fact was a amusement account it. Look advanced to more added agreeable from you! However, how can we communicate?
I used to be able to find good advice from your blog articles.
I do agree with all of the ideas you have presented in your post. They’re very convincing and will certainly work. Still, the posts are very short for beginners. Could you please extend them a little from next time? Thanks for the post.
Such a beneficial resource.
More articles like this would make the blogosphere richer.
I really admired the way this was presented.
The depth in this piece is commendable.
I’ll gladly bookmark this page.
I gained useful knowledge from this.
Thanks for creating this. It’s well done.
Aw, this was an incredibly nice post. Taking a few minutes and actual effort to produce a very good article… but what can I say… I put things off a lot and never seem to get anything done.
The depth in this write-up is noteworthy.
I’ll definitely be back for more.
Such a practical read.
Thanks for posting. It’s top quality.
I discovered useful points from this.
I gained useful knowledge from this.
Thanks for putting this up. It’s excellent.
Excellent post. I used to be checking constantly this blog and I am impressed! Very useful info specifically the last phase 🙂 I deal with such info a lot. I was looking for this particular information for a long time. Thank you and best of luck.
Spot on with this write-up, I really think this website needs much more consideration. I?ll most likely be once more to learn rather more, thanks for that info.
hey there and thanks to your info ? I have definitely picked up anything new from right here. I did then again expertise several technical points using this website, as I skilled to reload the website lots of instances prior to I may just get it to load properly. I were wondering in case your hosting is OK? No longer that I am complaining, but sluggish loading instances times will sometimes have an effect on your placement in google and could harm your high-quality score if ads and ***********|advertising|advertising|advertising and *********** with Adwords. Anyway I am adding this RSS to my email and could glance out for much more of your respective intriguing content. Ensure that you replace this again very soon..
Thanks for publishing. It’s well done.
I found new insight from this.
You have made some really good points there. I checked on the web to find out more about the issue and found most people will go along with your views on this site.
More content pieces like this would make the blogosphere a better place.
The depth in this content is exceptional.
Hello there! This blog post couldn’t be written any better! Going through this post reminds me of my previous roommate! He continually kept preaching about this. I’ll send this information to him. Fairly certain he will have a great read. Thank you for sharing!
Terrific work! This is the type of info that are supposed to be shared across the internet. Shame on Google for not positioning this publish upper! Come on over and discuss with my web site . Thank you =)
Excellent write-up. I definitely love this website. Stick with it!
That is a good tip particularly to those new to the blogosphere. Short but very precise information… Thanks for sharing this one. A must read article!
Howdy just wanted to give you a quick heads up. The text in your post seem to be running off the screen in Opera. I’m not sure if this is a format issue or something to do with web browser compatibility but I thought I’d post to let you know. The style and design look great though! Hope you get the issue solved soon. Cheers
Good day! I know this is kinda off topic but I’d figured I’d ask. Would you be interested in exchanging links or maybe guest authoring a blog post or vice-versa? My website discusses a lot of the same topics as yours and I feel we could greatly benefit from each other. If you are interested feel free to shoot me an e-mail. I look forward to hearing from you! Excellent blog by the way!
You need to be a part of a contest for one of the best websites online. I am going to highly recommend this web site!
Excellent weblog here! Additionally your site quite a bit up very fast! What web host are you the usage of? Can I am getting your associate hyperlink on your host? I want my site loaded up as fast as yours lol
I will also like to convey that most people who find themselves with out health insurance are usually students, self-employed and those that are jobless. More than half on the uninsured are really under the age of 35. They do not feel they are in need of health insurance as they are young along with healthy. Their own income is typically spent on houses, food, and also entertainment. Most people that do go to work either entire or not professional are not given insurance by way of their work so they move without due to the rising tariff of health insurance in the country. Thanks for the concepts you share through this website.
Simply want to say your article is as amazing. The clarity in your post is simply great and i can assume you are an expert on this subject. Well with your permission allow me to grab your RSS feed to keep updated with forthcoming post. Thanks a million and please carry on the gratifying work.
What?s Taking place i’m new to this, I stumbled upon this I’ve discovered It positively useful and it has aided me out loads. I’m hoping to give a contribution & assist different users like its helped me. Great job.
Hey very cool blog!! Man .. Beautiful .. Amazing .. I will bookmark your site and take the feeds also?I am happy to find so many useful information here in the post, we need develop more strategies in this regard, thanks for sharing. . . . . .
What i do not realize is in reality how you are now not really much more neatly-preferred than you may be now. You are so intelligent. You realize thus significantly relating to this matter, produced me individually believe it from a lot of numerous angles. Its like women and men are not involved except it is something to do with Woman gaga! Your individual stuffs excellent. At all times care for it up!
fantastic submit, very informative. I wonder why the other specialists of this sector do not notice this. You should continue your writing. I’m sure, you have a huge readers’ base already!
Hi, i think that i saw you visited my blog so i came to ?return the favor?.I am attempting to find things to improve my website!I suppose its ok to use some of your ideas!!
Woah! I’m really loving the template/theme of this blog. It’s simple, yet effective. A lot of times it’s difficult to get that “perfect balance” between user friendliness and visual appeal. I must say you have done a fantastic job with this. In addition, the blog loads very fast for me on Chrome. Exceptional Blog!
After research a couple of of the weblog posts on your website now, and I actually like your approach of blogging. I bookmarked it to my bookmark website checklist and will probably be checking again soon. Pls try my site as properly and let me know what you think.
Thanks for your article. I would also like to say that a health insurance brokerage also utilizes the benefit of the coordinators of your group insurance policy. The health insurance agent is given a listing of benefits looked for by somebody or a group coordinator. Such a broker can is find individuals or even coordinators that best fit those requirements. Then he gifts his advice and if the two of you agree, the particular broker formulates a legal contract between the two parties.
More articles like this would make the online space better.
I?m now not positive where you’re getting your info, but great topic. I must spend some time studying more or working out more. Thank you for fantastic info I was looking for this info for my mission.
Thank you a lot for sharing this with all people you really know what you are talking about! Bookmarked. Please also discuss with my website =). We may have a hyperlink alternate arrangement between us!
Thanks for putting this up. It’s brilliant work.
I absolutely valued the manner this was explained.
I took away a great deal from this.
I took away a great deal from this.
More blogs like this would make the blogosphere a better place.
More content pieces like this would make the internet richer.
Thanks for your post on the vacation industry. I’d also like contribute that if you’re a senior considering traveling, it’s absolutely vital that you buy travel cover for elderly people. When traveling, retirees are at high risk of getting a healthcare emergency. Getting the right insurance policies package in your age group can look after your health and provide you with peace of mind.
Thank you for the sensible critique. Me and my neighbor were just preparing to do a little research about this. We got a grab a book from our local library but I think I learned more clear from this post. I’m very glad to see such great info being shared freely out there.
Hiya! I know this is kinda off topic however , I’d figured I’d ask. Would you be interested in trading links or maybe guest authoring a blog article or vice-versa? My blog covers a lot of the same topics as yours and I feel we could greatly benefit from each other. If you might be interested feel free to send me an email. I look forward to hearing from you! Great blog by the way!
More articles like this would make the online space more useful.
More blogs like this would make the web more useful.
Thanks for publishing. It’s excellent.
I discovered useful points from this.
I’ll gladly bookmark this page.
I gained useful knowledge from this.
I genuinely valued the manner this was explained.
This submission is brilliant.
The detail in this content is remarkable.
More blogs like this would make the web a better place.
I gained useful knowledge from this.
Thanks for your suggestions. One thing I’ve got noticed is the fact banks as well as financial institutions understand the spending behaviors of consumers and also understand that the majority of people max outside their credit cards around the trips. They correctly take advantage of this kind of fact and start flooding your inbox as well as snail-mail box by using hundreds of Zero APR credit card offers just after the holiday season finishes. Knowing that should you be like 98 of American open public, you’ll soar at the possible opportunity to consolidate consumer credit card debt and switch balances towards 0 annual percentage rates credit cards.
The thoroughness in this content is commendable.
I truly enjoyed the way this was written.
I genuinely admired the way this was presented.
I took away a great deal from this.
I really valued the way this was presented.
More content pieces like this would make the web more useful.
More blogs like this would make the blogosphere more useful.
I truly liked the manner this was laid out.
I absolutely valued the way this was written.
The clarity in this article is remarkable.
The depth in this article is exceptional.
Unquestionably consider that that you said. Your favorite reason seemed to be on the net the simplest factor to take into account of. I say to you, I certainly get irked while other folks consider concerns that they just don’t understand about. You controlled to hit the nail upon the top as neatly as defined out the whole thing without having side effect , other folks could take a signal. Will likely be again to get more. Thanks
fantastic points altogether, you simply gained a new reader. What would you suggest in regards to your post that you made some days ago? Any positive?
This post is brilliant.
very good publish, i definitely love this website, carry on it
The detail in this write-up is exceptional.
Thanks for sharing. It’s top quality.
I particularly appreciated the way this was explained.
I do love the way you have framed this specific problem and it really does present me personally some fodder for consideration. However, coming from everything that I have seen, I simply hope when the actual feedback pile on that people today remain on issue and in no way embark on a soap box of the news du jour. Yet, thank you for this superb point and while I do not agree with the idea in totality, I respect your viewpoint.
Thanks for your article. I would like to say that a health insurance brokerage also works for the benefit of the actual coordinators of the group insurance cover. The health broker is given a summary of benefits desired by individuals or a group coordinator. Such a broker does indeed is search for individuals or coordinators which in turn best go with those desires. Then he offers his tips and if each party agree, the actual broker formulates a binding agreement between the two parties.
More posts like this would make the web more useful.
More posts like this would make the web a better place.
This is the kind of information I truly appreciate.
There are some fascinating points in time in this article but I don?t know if I see all of them heart to heart. There may be some validity but I will take maintain opinion till I look into it further. Good article , thanks and we wish extra! Added to FeedBurner as effectively
I discovered useful points from this.
Such a helpful insight.
I absolutely liked the way this was laid out.
Such a valuable read.
Such a beneficial bit of content.
Thanks for publishing. It’s brilliant work.
I’ll definitely be back for more.
I took away a great deal from this.
The breadth in this article is praiseworthy.
Such a valuable resource.
The thoroughness in this piece is noteworthy.
Can I simply say what a reduction to find someone who really knows what theyre speaking about on the internet. You undoubtedly know tips on how to convey a problem to light and make it important. More folks have to learn this and perceive this facet of the story. I cant consider youre not more popular since you undoubtedly have the gift.
It’s nearly impossible to find educated people on this topic, however, you seem like you know what you’re talking about! Thanks
Hey there! This is my first comment here so I just wanted to give a quick shout out and tell you I truly enjoy reading through your posts. Can you recommend any other blogs/websites/forums that deal with the same topics? Thank you!
I?m not sure where you are getting your information, but great topic. I needs to spend some time learning more or understanding more. Thanks for magnificent info I was looking for this information for my mission.
An impressive share, I simply given this onto a colleague who was doing a bit evaluation on this. And he in reality bought me breakfast as a result of I found it for him.. smile. So let me reword that: Thnx for the treat! However yeah Thnkx for spending the time to discuss this, I feel strongly about it and love studying more on this topic. If doable, as you turn out to be expertise, would you mind updating your blog with more particulars? It’s highly helpful for me. Huge thumb up for this weblog put up!
More blogs like this would make the web a better place.
This is the kind of content I value most.
I learned a lot from this.
I really liked the manner this was written.
I discovered useful points from this.
I’ll certainly return to read more.
Such a practical read.
I gained useful knowledge from this.
I gained useful knowledge from this.
Such a useful insight.
More articles like this would make the online space a better place.
I took away a great deal from this.
I truly appreciated the style this was presented.
I took away a great deal from this.
I just added this website to my feed reader, excellent stuff. Cannot get enough!
Thanks for your write-up. One other thing is that if you are selling your property yourself, one of the challenges you need to be mindful of upfront is when to deal with property inspection accounts. As a FSBO retailer, the key to successfully moving your property and also saving money in real estate agent income is knowledge. The more you already know, the more stable your property sales effort are going to be. One area that this is particularly crucial is information about home inspections.
I?ll immediately seize your rss as I can’t to find your e-mail subscription link or e-newsletter service. Do you have any? Kindly permit me understand so that I may just subscribe. Thanks.
I took away a great deal from this.
This piece is incredible.
You’ve evidently researched well.
The depth in this content is exceptional.
Such a useful resource.
Thanks for creating this. It’s brilliant work.
I found new insight from this.
Another thing I have noticed is that often for many people, below-average credit is the reaction of circumstances above their control. As an example they may be actually saddled through an illness so they really have higher bills for collections. It would be due to a employment loss and the inability to work. Sometimes divorce proceedings can really send the financial circumstances in the undesired direction. Many thanks for sharing your opinions on this weblog.
I discovered useful points from this.
I just added this website to my google reader, great stuff. Cannot get enough!
More blogs like this would make the blogosphere more useful.
Such a valuable insight.
I really valued the manner this was laid out.
Such a helpful insight.
It’s the best time to make some plans for the future and it’s time to be happy. I have read this post and if I could I desire to suggest you few interesting things or advice. Maybe you could write next articles referring to this article. I wish to read even more things about it!
You’ve obviously spent time crafting this.
I’ll gladly bookmark this page.
I found new insight from this.
You’ve evidently spent time crafting this.
I discovered useful points from this.
I really valued the approach this was explained.
Such a practical resource.
Greetings! Very useful advice in this particular post! It is the little changes which will make the most significant changes. Many thanks for sharing!
This post is incredible.
Spot on with this write-up, I truly assume this website wants much more consideration. I?ll in all probability be once more to learn way more, thanks for that info.
very good submit, i definitely love this web site, carry on it
I have discovered some new points from your web-site about personal computers. Another thing I’ve always thought is that laptop computers have become something that each home must have for many reasons. They provide convenient ways in which to organize households, pay bills, shop, study, tune in to music and in some cases watch tv series. An innovative strategy to complete all of these tasks is a notebook computer. These computer systems are portable ones, small, highly effective and mobile.
Attractive portion of content. I just stumbled upon your web site and in accession capital to assert that I get in fact loved account your weblog posts. Anyway I?ll be subscribing on your augment and even I success you get right of entry to persistently quickly.
I genuinely admired the style this was laid out.
Thanks for posting. It’s brilliant work.
This post is well-written.
I’ll surely bookmark this page.
Right here is the perfect site for anyone who really wants to find out about this topic. You understand so much its almost hard to argue with you (not that I really would want to…HaHa). You definitely put a fresh spin on a topic which has been discussed for ages. Excellent stuff, just wonderful.
I truly appreciated the style this was explained.
I discovered useful points from this.
The depth in this write-up is exceptional.
Good day! This post could not be written any better! Reading through this post reminds me of my old room mate! He always kept talking about this. I will forward this page to him. Pretty sure he will have a good read. Thank you for sharing!
This is really interesting, You are a very skilled blogger. I have joined your feed and look forward to seeking more of your excellent post. Also, I have shared your website in my social networks!
Hi, I do think this is an excellent site. I stumbledupon it 😉 I will come back yet again since i have bookmarked it. Money and freedom is the greatest way to change, may you be rich and continue to guide other people.
This is a topic that is near to my heart… Thank you! Exactly where can I find the contact details for questions?
This is the kind of information I value most.
This is the kind of content I enjoy reading.
Such a informative read.
Such a useful bit of content.
I gained useful knowledge from this.
Such a practical bit of content.
Greetings! Very helpful advice in this particular article! It’s the little changes that will make the greatest changes. Thanks for sharing!
Such a informative resource.
One thing I would like to say is that before acquiring more laptop memory, look at the machine within which it could be installed. When the machine is definitely running Windows XP, for instance, the particular memory threshold is 3.25GB. Putting in over this would merely constitute some sort of waste. Make sure one’s mother board can handle the actual upgrade quantity, as well. Interesting blog post.
Do you mind if I quote a couple of your posts as long as I provide credit and sources back to your weblog? My website is in the exact same area of interest as yours and my users would certainly benefit from a lot of the information you present here. Please let me know if this okay with you. Thank you!
Such a informative bit of content.
More posts like this would make the web richer.
More posts like this would make the web more useful.
Such a useful insight.
I found new insight from this.
I truly liked the manner this was laid out.
More articles like this would make the blogosphere better.
May I simply just say what a comfort to uncover a person that actually knows what they’re talking about over the internet. You actually know how to bring an issue to light and make it important. More and more people should check this out and understand this side of the story. I was surprised you’re not more popular because you definitely have the gift.
I really admired the style this was laid out.
I discovered useful points from this.
Such a helpful bit of content.
Thanks for creating this. It’s top quality.
I particularly liked the style this was explained.
More posts like this would make the blogosphere more useful.
I’ll definitely bookmark this page.
This submission is valuable.
Excellent post! We are linking to this particularly great article on our website. Keep up the great writing.
I just couldn’t depart your website before suggesting that I extremely enjoyed the standard info a person provide for your visitors? Is going to be back often in order to check up on new posts
I truly admired the approach this was laid out.
You’ve undoubtedly done your homework.
Such a useful read.
Thanks for putting this up. It’s excellent.
More content pieces like this would make the online space a better place.
Thanks for posting. It’s well done.
I truly admired the approach this was presented.
More posts like this would make the web more useful.
Such a valuable read.
Thanks for your post. I also believe that laptop computers are getting to be more and more popular nowadays, and now are sometimes the only kind of computer used in a household. The reason is that at the same time that they’re becoming more and more inexpensive, their working power keeps growing to the point where they are as highly effective as desktop computers out of just a few years back.
Thanks for creating this. It’s top quality.
Such a helpful resource.
The thoroughness in this piece is exceptional.
The thoroughness in this article is commendable.
Thanks for sharing. It’s a solid effort.
I really admired the approach this was written.
Thanks for sharing. It’s a solid effort.
The detail in this piece is commendable.
Such a useful bit of content.
I found new insight from this.
Such a practical insight.
Thanks for sharing. It’s well done.
Thanks for your posting. My partner and i have generally seen that almost all people are needing to lose weight because they wish to show up slim in addition to looking attractive. However, they do not generally realize that there are additional benefits for you to losing weight additionally. Doctors say that obese people are afflicted by a variety of ailments that can be directly attributed to their excess weight. Fortunately that people that are overweight in addition to suffering from a variety of diseases are able to reduce the severity of their own illnesses by way of losing weight. You are able to see a continuous but noticeable improvement in health if even a small amount of weight loss is accomplished.
Everything is very open with a very clear clarification of the challenges. It was really informative. Your website is useful. Thanks for sharing!
I gained useful knowledge from this.
I discovered useful points from this.
I absolutely liked the way this was laid out.
This write-up is amazing.
This is the kind of post I truly appreciate.
More blogs like this would make the blogosphere better.
This submission is well-written.
You have made some good points there. I looked on the web for additional information about the issue and found most people will go along with your views on this website.
Hey very cool website!! Man .. Beautiful .. Amazing .. I will bookmark your web site and take the feeds also?I am happy to find a lot of useful information here in the post, we need work out more techniques in this regard, thanks for sharing. . . . . .
Fantastic goods from you, man. I have understand your stuff previous to and you are just too excellent. I actually like what you’ve acquired here, really like what you’re stating and the way in which you say it. You make it entertaining and you still take care of to keep it wise. I can’t wait to read far more from you. This is really a great website.
More articles like this would make the internet better.
Thank you for another informative blog. Where else could I get that type of info written in such a perfect way? I have a project that I’m just now working on, and I’ve been on the look out for such information.
I am no longer certain the place you’re getting your information, however good topic. I needs to spend a while studying more or figuring out more. Thank you for fantastic information I was searching for this information for my mission.
I’ll surely return to read more.
I can’t express how much I value the effort the author has put into writing this outstanding piece of content. The clarity of the writing, the depth of analysis, and the abundance of information provided are simply impressive. Her passion for the subject is apparent, and it has certainly resonated with me. Thank you, author, for offering your wisdom and enhancing our lives with this exceptional article!
Have you ever considered writing an ebook or guest authoring on other sites? I have a blog based upon on the same topics you discuss and would love to have you share some stories/information. I know my audience would value your work. If you are even remotely interested, feel free to shoot me an e mail.
very good submit, i actually love this website, keep on it
I am really impressed with your writing talents and also with the structure to your blog. Is this a paid topic or did you modify it yourself? Anyway stay up the nice high quality writing, it is uncommon to peer a great weblog like this one these days..
Way cool! Some extremely valid points! I appreciate you writing this write-up plus the rest of the site is extremely good.
There is definately a great deal to know about this issue. I really like all of the points you have made.
There are certainly lots of details like that to take into consideration. That could be a great point to deliver up. I supply the thoughts above as general inspiration however clearly there are questions just like the one you deliver up where an important thing can be working in trustworthy good faith. I don?t know if best practices have emerged around things like that, but I am positive that your job is clearly identified as a good game. Both boys and girls feel the impact of just a moment?s pleasure, for the remainder of their lives.
Hi there, simply turned into alert to your weblog thru Google, and found that it’s really informative. I am going to be careful for brussels. I will appreciate should you proceed this in future. Lots of other folks will likely be benefited out of your writing. Cheers!
Thanks for your posting. What I want to say is that when looking for a good on the web electronics shop, look for a internet site with entire information on critical indicators such as the level of privacy statement, safety measures details, any payment procedures, and other terms plus policies. Usually take time to read the help and FAQ pieces to get a far better idea of what sort of shop functions, what they are capable of doing for you, and the way you can maximize the features.
This website is known as a walk-by means of for all the info you wanted about this and didn?t know who to ask. Glimpse here, and also you?ll positively uncover it.
Hey! This is kind of off topic but I need some guidance from an established blog. Is it tough to set up your own blog? I’m not very techincal but I can figure things out pretty quick. I’m thinking about setting up my own but I’m not sure where to begin. Do you have any points or suggestions? Thanks
When I originally commented I clicked the “Notify me when new comments are added” checkbox and now each time a comment is added I get three emails with the same comment. Is there any way you can remove people from that service? Bless you!
I have noticed that online degree is getting popular because getting your college degree online has developed into a popular selection for many people. Many people have not had an opportunity to attend a traditional college or university yet seek the increased earning possibilities and a better job that a Bachelor Degree offers. Still some others might have a degree in one discipline but want to pursue something they now develop an interest in.
Right here is the right website for anyone who wishes to understand this topic. You realize a whole lot its almost hard to argue with you (not that I personally would want to…HaHa). You definitely put a brand new spin on a topic that’s been written about for a long time. Excellent stuff, just excellent.
Having read this I believed it was rather informative. I appreciate you finding the time and effort to put this content together. I once again find myself personally spending way too much time both reading and leaving comments. But so what, it was still worthwhile!
I’m very happy to read this. This is the type of manual that needs to be given and not the random misinformation that is at the other blogs. Appreciate your sharing this best doc.
What i don’t realize is in truth how you are no longer actually much more smartly-appreciated than you might be right now. You’re so intelligent. You understand thus significantly in terms of this topic, made me for my part believe it from so many numerous angles. Its like men and women are not fascinated until it?s one thing to do with Girl gaga! Your individual stuffs nice. At all times maintain it up!
Thanks for your publication. I would also like to say this that the very first thing you will need to complete is find out if you really need repairing credit. To do that you need to get your hands on a copy of your credit rating. That should not be difficult, since the government makes it necessary that you are allowed to have one no cost copy of your real credit report yearly. You just have to ask the right individuals. You can either read the website for that Federal Trade Commission or even contact one of the main credit agencies specifically.
Thank you for the auspicious writeup. It in fact was a amusement account it. Look advanced to far added agreeable from you! By the way, how could we communicate?
I seriously love your blog.. Great colors & theme. Did you make this web site yourself? Please reply back as I’m trying to create my very own site and would like to learn where you got this from or exactly what the theme is named. Many thanks!
I was able to find good info from your content.
Spot on with this write-up, I actually think this web site wants rather more consideration. I?ll in all probability be once more to learn rather more, thanks for that info.
Thanks for your posting on the travel industry. I’d also like to add that if you’re a senior contemplating traveling, it is absolutely vital that you buy travel cover for golden-agers. When traveling, senior citizens are at high risk of experiencing a healthcare emergency. Buying the right insurance cover package in your age group can safeguard your health and provide you with peace of mind.
Thanks for your posting. I also believe laptop computers are getting to be more and more popular right now, and now will often be the only kind of computer utilized in a household. The reason being at the same time they are becoming more and more economical, their computing power keeps growing to the point where these are as powerful as personal computers through just a few years back.
I couldn’t refrain from commenting. Very well written.
Whoa! This blog looks exactly like my old one! It’s on a totally different topic but it has pretty much the same layout and design. Wonderful choice of colors!
Aw, this was an incredibly good post. Taking a few minutes and actual effort to make a good article… but what can I say… I put things off a whole lot and never manage to get nearly anything done.
Interesting article. It’s very unfortunate that over the last ten years, the travel industry has had to tackle terrorism, SARS, tsunamis, flu virus, swine flu, along with the first ever real global downturn. Through all this the industry has really proven to be strong, resilient along with dynamic, obtaining new approaches to deal with adversity. There are continually fresh challenges and opportunity to which the field must just as before adapt and react.
One thing I’d like to say is that before obtaining more computer system memory, look into the machine in which it would be installed. If the machine is actually running Windows XP, for instance, the particular memory limit is 3.25GB. Applying a lot more than this would merely constitute a waste. Make certain that one’s motherboard can handle an upgrade quantity, as well. Thanks for your blog post.
Very good article. I am dealing with some of these issues as well..
Very good info. Lucky me I recently found your blog by accident (stumbleupon). I’ve saved it for later.
Thanks for every other informative site. Where else may I get that type of info written in such an ideal manner? I’ve a project that I’m simply now operating on, and I’ve been on the look out for such information.
whoah this blog is great i love reading your articles. Keep up the good work! You know, lots of people are looking around for this info, you can help them greatly.
Very good article. I will be facing many of these issues as well..
You’ve made some decent points there. I looked on the internet to find out more about the issue and found most people will go along with your views on this website.
You actually make it seem so easy with your presentation but I find this matter to be actually something which I think I would never understand. It seems too complex and very broad for me. I am looking forward for your next post, I will try to get the hang of it!
This is a topic that’s near to my heart… Many thanks! Where are your contact details though?
Saved as a favorite, I like your site.
Hello there! I simply would like to offer you a huge thumbs up for the excellent info you have got right here on this post. I will be coming back to your site for more soon.
I used to be able to find good info from your articles.
Hello, I think your website could be having web browser compatibility issues. Whenever I look at your blog in Safari, it looks fine however when opening in I.E., it’s got some overlapping issues. I just wanted to give you a quick heads up! Other than that, fantastic website!
Hello my friend! I want to say that this article is awesome, nice written and include approximately all significant infos. I¦d like to look extra posts like this .
Also a thing to mention is that an online business administration training is designed for individuals to be able to smoothly proceed to bachelors degree education. The Ninety credit education meets the other bachelor diploma requirements then when you earn your associate of arts in BA online, you’ll have access to the newest technologies with this field. Some reasons why students would like to get their associate degree in business is because they are interested in the field and want to obtain the general education and learning necessary previous to jumping right bachelor education program. Thanks alot : ) for the tips you actually provide in your blog.
May I simply just say what a comfort to discover someone that truly understands what they are talking about online. You definitely know how to bring an issue to light and make it important. A lot more people need to check this out and understand this side of the story. I was surprised that you’re not more popular since you certainly have the gift.
I believe that avoiding ready-made foods is a first step for you to lose weight. They might taste great, but packaged foods possess very little nutritional value, making you consume more only to have enough power to get throughout the day. If you are constantly ingesting these foods, changing to grain and other complex carbohydrates will make you to have more vigor while ingesting less. Great blog post.
Hello! I simply want to give you a big thumbs up for the excellent info you have got here on this post. I am returning to your website for more soon.
Pretty! This was an extremely wonderful article. Many thanks for providing this info.
I’m not sure exactly why but this web site is loading incredibly slow for me. Is anyone else having this problem or is it a issue on my end? I’ll check back later and see if the problem still exists.
That is the proper blog for anyone who needs to seek out out about this topic. You realize a lot its virtually hard to argue with you (not that I truly would need?HaHa). You definitely put a brand new spin on a subject thats been written about for years. Nice stuff, simply nice!
Good post. I study something tougher on completely different blogs everyday. It should all the time be stimulating to learn content material from different writers and follow a bit something from their store. I?d prefer to make use of some with the content on my blog whether or not you don?t mind. Natually I?ll give you a hyperlink on your net blog. Thanks for sharing.
You need to take part in a contest for one of the greatest blogs on the internet. I will highly recommend this web site!
This is a really good tip particularly to those new to the blogosphere. Brief but very precise information… Thanks for sharing this one. A must read post!
What i do not realize is actually how you are not really much more well-liked than you might be now. You are very intelligent. You realize therefore considerably relating to this subject, made me personally consider it from so many varied angles. Its like men and women aren’t fascinated unless it is one thing to accomplish with Lady gaga! Your own stuffs excellent. Always maintain it up!
I’m amazed, I have to admit. Rarely do I come across a blog that’s equally educative and interesting, and let me tell you, you have hit the nail on the head. The issue is something that too few people are speaking intelligently about. I’m very happy I came across this during my search for something concerning this.
I have seen that today, more and more people will be attracted to digital cameras and the subject of digital photography. However, to be a photographer, you have to first shell out so much of your time deciding the exact model of photographic camera to buy plus moving out of store to store just so you can buy the least expensive camera of the trademark you have decided to settle on. But it would not end right now there. You also have to consider whether you should buy a digital video camera extended warranty. Many thanks for the good points I accumulated from your blog.
This is a terrific web site, could you be involved in doing an interview about how you developed it? If so e-mail me!
This is a topic that’s near to my heart… Take care! Exactly where are your contact details though?
Greetings, I do believe your site could be having web browser compatibility issues. When I look at your website in Safari, it looks fine however, when opening in I.E., it’s got some overlapping issues. I merely wanted to provide you with a quick heads up! Other than that, great site.
This submission is outstanding.
Great information. Lucky me I recently found your site by accident (stumbleupon). I’ve book marked it for later!
That is a good tip especially to those new to the blogosphere. Brief but very accurate information… Thanks for sharing this one. A must read post!
Good info. Lucky me I came across your website by accident (stumbleupon). I’ve saved it for later!
I seriously love your site.. Very nice colors & theme. Did you build this amazing site yourself? Please reply back as I’m trying to create my very own site and would love to know where you got this from or what the theme is named. Cheers.
It’s hard to come by experienced people for this topic, but you sound like you know what you’re talking about! Thanks
Way cool! Some very valid points! I appreciate you penning this article and also the rest of the website is extremely good.
This is the kind of information I truly appreciate.
Greetings! I know this is kinda off topic but I was wondering which blog platform are you using for this site? I’m getting sick and tired of WordPress because I’ve had issues with hackers and I’m looking at options for another platform. I would be fantastic if you could point me in the direction of a good platform.
Hi there! This post could not be written any better! Looking at this post reminds me of my previous roommate! He always kept preaching about this. I’ll forward this information to him. Fairly certain he will have a great read. Thanks for sharing!
Thanks for the write-up. I have generally seen that a lot of people are wanting to lose weight simply because wish to look slim as well as attractive. However, they do not continually realize that there are additional benefits just for losing weight additionally. Doctors declare that over weight people have problems with a variety of ailments that can be perfectely attributed to the excess weight. Fortunately that people who definitely are overweight and also suffering from numerous diseases can help to eliminate the severity of their illnesses by way of losing weight. It’s possible to see a gradual but marked improvement in health as soon as even a minor amount of fat loss is realized.
You’re so interesting! I do not believe I have read through something like that before. So nice to find somebody with original thoughts on this issue. Seriously.. many thanks for starting this up. This website is something that’s needed on the web, someone with a little originality.
There is noticeably a bundle to find out about this. I assume you made certain good points in features also.
Wonderful article! We are linking to this great post on our site. Keep up the great writing.
I have observed that over the course of constructing a relationship with real estate managers, you’ll be able to come to understand that, in most real estate financial transaction, a commission is paid. All things considered, FSBO sellers never “save” the percentage. Rather, they try to win the commission simply by doing a agent’s job. In completing this task, they invest their money plus time to conduct, as best they’re able to, the obligations of an adviser. Those jobs include displaying the home through marketing, representing the home to buyers, creating a sense of buyer emergency in order to prompt an offer, preparing home inspections, handling qualification inspections with the bank, supervising repairs, and assisting the closing of the deal.
After I originally commented I seem to have clicked the -Notify me when new comments are added- checkbox and now every time a comment is added I recieve 4 emails with the same comment. Perhaps there is an easy method you can remove me from that service? Thanks.
Can I simply say what a relief to find a person that genuinely knows what they are talking about on the web. You actually realize how to bring an issue to light and make it important. More and more people must read this and understand this side of the story. It’s surprising you are not more popular because you certainly have the gift.
Nice post. I learn something totally new and challenging on websites I stumbleupon everyday. It’s always useful to read articles from other writers and use a little something from their sites.
It’s hard to come by educated people about this subject, however, you sound like you know what you’re talking about! Thanks
Pretty! This has been an incredibly wonderful post. Thanks for providing this info.
Excellent write-up. I absolutely appreciate this site. Continue the good work!
Thanks for helping me to acquire new concepts about pcs. I also have belief that certain of the best ways to maintain your notebook computer in excellent condition is to use a hard plastic case, and also shell, that suits over the top of one’s computer. Most of these protective gear are generally model distinct since they are made to fit perfectly above the natural covering. You can buy these directly from owner, or from third party sources if they are designed for your notebook, however not all laptop can have a covering on the market. Again, thanks for your guidelines.
Nice post. I learn something totally new and challenging on blogs I stumbleupon on a daily basis. It will always be interesting to read through content from other writers and practice something from their sites.
The next time I read a blog, I hope that it won’t disappoint me just as much as this particular one. I mean, Yes, it was my choice to read through, but I truly thought you’d have something interesting to talk about. All I hear is a bunch of whining about something you could fix if you were not too busy looking for attention.
Howdy! I could have sworn I’ve visited this blog before but after going through a few of the posts I realized it’s new to me. Anyways, I’m certainly delighted I stumbled upon it and I’ll be bookmarking it and checking back frequently.
I like it whenever people come together and share views. Great site, continue the good work.
Good post. I will be facing some of these issues as well..
Aw, this was an extremely good post. Spending some time and actual effort to make a superb article… but what can I say… I hesitate a lot and don’t seem to get nearly anything done.
I’m really enjoying the design and layout of your blog. It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often. Did you hire out a designer to create your theme? Fantastic work!
Aw, this was an exceptionally nice post. Taking a few minutes and actual effort to make a top notch article… but what can I say… I hesitate a whole lot and don’t manage to get nearly anything done.
You are so cool! I don’t believe I have read anything like this before. So good to find somebody with some unique thoughts on this topic. Seriously.. thanks for starting this up. This web site is one thing that is needed on the internet, someone with a bit of originality.
I could not refrain from commenting. Well written.
You need to be a part of a contest for one of the best sites on the net. I most certainly will recommend this blog!
You are so cool! I do not think I have read through something like this before. So nice to find someone with some original thoughts on this subject. Really.. thanks for starting this up. This site is one thing that is needed on the internet, someone with some originality.
Very good article. I absolutely appreciate this website. Thanks!
My programmer is trying to persuade me to move to .net from PHP. I have always disliked the idea because of the costs. But he’s tryiong none the less. I’ve been using WordPress on various websites for about a year and am anxious about switching to another platform. I have heard excellent things about blogengine.net. Is there a way I can import all my wordpress posts into it? Any kind of help would be really appreciated!
The next time I read a blog, I hope that it doesnt disappoint me as much as this one. I mean, I do know it was my choice to learn, but I truly thought youd have one thing fascinating to say. All I hear is a bunch of whining about one thing that you possibly can fix in case you werent too busy looking for attention.
Одним из ключевых аспектов ОЗДС является также обучение персонала, ответственного за борьбу с дератами, в правилах безопасности, применении различных методов и технологий, а также в мониторинге и контроле за эффективностью применяемых мероприятий.
Биологические методы также могут быть включены в состав ОЗДС, что делает их еще более экологически безопасными. Это может включать использование препаратов на основе микроорганизмов или хищных насекомых, которые питаются дератами или их личинками, контролируя их популяцию естественным путем.
whoah this weblog is fantastic i really like studying your articles. Stay up the good work! You already know, many persons are looking around for this information, you can aid them greatly.
Such a valuable read.
I can’t believe how amazing this article is! The author has done a fantastic job of presenting the information in an engaging and informative manner. I can’t thank him enough for sharing such valuable insights that have definitely enriched my awareness in this topic. Kudos to him for producing such a masterpiece!
This piece is insightful.
Such a useful read.
Hi are using WordPress for your site platform? I’m new to the blog world but I’m trying to get started and create my own. Do you need any coding expertise to make your own blog? Any help would be really appreciated!
always i used to read smaller content which also clear their motive, and that is also happening with this paragraph which I am reading now.
I love your blog.. very nice colors & theme. Did you create this website yourself or did you hire someone to do it for you? Plz reply as I’m looking to design my own blog and would like to know where u got this from. thanks a lot
Such a practical insight.
Hi there! This article couldn’t be written any better! Looking through this article reminds me of my previous roommate! He continually kept preaching about this. I most certainly will send this information to him. Pretty sure he’ll have a very good read. I appreciate you for sharing!
This is a topic that is near to my heart… Cheers! Where can I find the contact details for questions?
I must thank you for the efforts you have put in writing this blog. I really hope to check out the same high-grade blog posts by you in the future as well. In fact, your creative writing abilities has motivated me to get my very own site now 😉
I needed to thank you for this good read!! I absolutely loved every little bit of it. I have you book-marked to check out new things you post…
An impressive share! I’ve just forwarded this onto a co-worker who was conducting a little research on this. And he actually ordered me breakfast because I stumbled upon it for him… lol. So let me reword this…. Thank YOU for the meal!! But yeah, thanks for spending the time to discuss this matter here on your internet site.
Howdy! I could have sworn I’ve visited this website before but after browsing through some of the articles I realized it’s new to me. Anyhow, I’m definitely pleased I stumbled upon it and I’ll be book-marking it and checking back often.
Good article. I’m experiencing many of these issues as well..
This blog was… how do you say it? Relevant!! Finally I have found something which helped me. Appreciate it!
Do you have a spam problem on this blog; I also am a blogger, and I was wondering your situation; we have developed some nice practices and we are looking to trade methods with others, please shoot me an email if interested.
Thank you for this article. I will also like to express that it can possibly be hard if you find yourself in school and just starting out to initiate a long credit score. There are many pupils who are simply just trying to endure and have a long or positive credit history are often a difficult issue to have.
We absolutely love your blog and find nearly all of your post’s to be just what I’m looking for. can you offer guest writers to write content for you? I wouldn’t mind composing a post or elaborating on some of the subjects you write concerning here. Again, awesome site!
Pretty! This has been an extremely wonderful post. Thanks for providing these details.
I’ll definitely bookmark this page.
Nice post. I learn something totally new and challenging on blogs I stumbleupon on a daily basis. It will always be exciting to read through content from other writers and use something from their websites.
Everything is very open with a precise clarification of the challenges. It was really informative. Your website is extremely helpful. Thanks for sharing.
Such a helpful bit of content.
Thanks for publishing. It’s a solid effort.
The thoroughness in this write-up is commendable.
I like looking through a post that can make people think. Also, thanks for allowing for me to comment.
I really love your website.. Excellent colors & theme. Did you make this website yourself? Please reply back as I’m looking to create my own personal website and would like to learn where you got this from or what the theme is called. Thanks!
Very good post. I’m experiencing many of these issues as well..
I love reading a post that will make men and women think. Also, thank you for allowing for me to comment.
I’m in awe of the author’s capability to make intricate concepts accessible to readers of all backgrounds. This article is a testament to her expertise and passion to providing helpful insights. Thank you, author, for creating such an engaging and insightful piece. It has been an unforgettable experience to read!
I?m impressed, I must say. Actually not often do I encounter a weblog that?s each educative and entertaining, and let me let you know, you’ve hit the nail on the head. Your concept is excellent; the issue is one thing that not enough people are talking intelligently about. I’m very comfortable that I stumbled across this in my seek for one thing regarding this.
Hello just wanted to give you a quick heads up. The text in your content seem to be running off the screen in Firefox. I’m not sure if this is a formatting issue or something to do with internet browser compatibility but I thought I’d post to let you know. The design look great though! Hope you get the issue solved soon. Kudos
I?m now not certain the place you are getting your information, however good topic. I needs to spend a while finding out more or understanding more. Thank you for excellent info I was looking for this info for my mission.
An impressive share! I have just forwarded this onto a friend who was conducting a little research on this. And he in fact ordered me lunch because I found it for him… lol. So allow me to reword this…. Thank YOU for the meal!! But yeah, thanx for spending time to discuss this matter here on your site.
Your style is very unique in comparison to other people I’ve read stuff from. Thank you for posting when you have the opportunity, Guess I will just bookmark this blog.
Good post. I learn something totally new and challenging on sites I stumbleupon on a daily basis. It will always be exciting to read through articles from other writers and use something from other sites.
Hi! I just would like to offer you a huge thumbs up for the great info you have got here on this post. I’ll be returning to your site for more soon.
I was able to find good advice from your content.
Your style is really unique compared to other people I’ve read stuff from. Thanks for posting when you’ve got the opportunity, Guess I’ll just book mark this site.
You have made some good points there. I looked on the internet to learn more about the issue and found most individuals will go along with your views on this site.
I used to be able to find good advice from your blog articles.
I’m amazed, I must say. Rarely do I come across a blog that’s both educative and engaging, and without a doubt, you have hit the nail on the head. The issue is something which too few men and women are speaking intelligently about. I am very happy that I stumbled across this in my search for something regarding this.
I think other website proprietors should take this website as an model, very clean and excellent user friendly style and design, as well as the content. You are an expert in this topic!
I have seen that nowadays, more and more people are attracted to surveillance cameras and the discipline of picture taking. However, to be a photographer, it’s important to first shell out so much time deciding the exact model of digital camera to buy and also moving store to store just so you might buy the least expensive camera of the brand you have decided to select. But it doesn’t end generally there. You also have to consider whether you can purchase a digital camera extended warranty. Many thanks for the good ideas I gained from your blog.
You ought to take part in a contest for one of the most useful blogs on the web. I most certainly will highly recommend this website!
I quite like looking through a post that can make men and women think. Also, many thanks for allowing me to comment.
After looking over a handful of the blog articles on your website, I really like your way of writing a blog. I saved as a favorite it to my bookmark site list and will be checking back in the near future. Take a look at my web site too and tell me your opinion.
That is a great tip especially to those new to the blogosphere. Brief but very precise information… Thank you for sharing this one. A must read article.
This site truly has all of the info I wanted about this subject and didn’t know who to ask.
I quite like reading through a post that will make men and women think. Also, many thanks for permitting me to comment.
Saved as a favorite, I love your site.
fantastic points altogether, you just gained a brand new reader. What would you recommend about your post that you made some days ago? Any positive?
Another thing I’ve really noticed is that often for many people, a bad credit score is the reaction to circumstances past their control. For instance they may have already been saddled through an illness so they have higher bills going to collections. It may be due to a occupation loss or even the inability to work. Sometimes divorce proceedings can send the money in the wrong direction. Many thanks sharing your opinions on this blog.
Spot on with this write-up, I honestly believe that this website needs a lot more attention. I’ll probably be returning to read through more, thanks for the information.
I couldn’t refrain from commenting. Very well written!
An interesting discussion is definitely worth comment. I do think that you ought to publish more on this topic, it may not be a taboo matter but typically people don’t speak about such issues. To the next! Many thanks!
Everyone loves it when people come together and share opinions. Great blog, continue the good work.
Your style is very unique in comparison to other folks I have read stuff from. Many thanks for posting when you have the opportunity, Guess I’ll just bookmark this site.
Way cool! Some extremely valid points! I appreciate you penning this post and also the rest of the website is extremely good.
Hi, I do think this is a great web site. I stumbledupon it 😉 I will come back once again since i have bookmarked it. Money and freedom is the best way to change, may you be rich and continue to guide others.
I blog often and I seriously appreciate your content. Your article has really peaked my interest. I’m going to book mark your blog and keep checking for new information about once per week. I subscribed to your Feed as well.
After I initially commented I appear to have clicked on the -Notify me when new comments are added- checkbox and from now on every time a comment is added I get 4 emails with the exact same comment. There has to be an easy method you are able to remove me from that service? Appreciate it.
Way cool! Some extremely valid points! I appreciate you penning this post plus the rest of the website is also very good.
When I initially commented I seem to have clicked the -Notify me when new comments are added- checkbox and now each time a comment is added I recieve 4 emails with the same comment. Is there a means you can remove me from that service? Many thanks.
Thanks for the ideas you have discussed here. Furthermore, I believe there are a few factors that really keep your automobile insurance premium straight down. One is, to take into consideration buying autos that are within the good set of car insurance companies. Cars which have been expensive will be more at risk of being snatched. Aside from that insurance coverage is also in line with the value of your vehicle, so the more costly it is, then the higher your premium you spend.
I acquired more new things on this fat loss issue. 1 issue is that good nutrition is very vital whenever dieting. A big reduction in bad foods, sugary foods, fried foods, sweet foods, pork, and white colored flour products can be necessary. Holding wastes harmful bacteria, and wastes may prevent desired goals for fat-loss. While particular drugs momentarily solve the problem, the awful side effects are certainly not worth it, they usually never offer more than a non permanent solution. This is a known incontrovertible fact that 95 of dietary fads fail. Many thanks sharing your ideas on this web site.
Thanks for the ideas you have shared here. One more thing I would like to express is that laptop memory needs generally increase along with other innovations in the technologies. For instance, whenever new generations of cpus are introduced to the market, there’s usually a similar increase in the type calls for of both computer system memory plus hard drive room. This is because the program operated through these processors will inevitably increase in power to make use of the new technological know-how.
Hey there are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and create my own. Do you need any coding expertise to make your own blog? Any help would be greatly appreciated!
Having read this I believed it was very enlightening. I appreciate you spending some time and energy to put this short article together. I once again find myself spending a significant amount of time both reading and commenting. But so what, it was still worthwhile!
Very good post. I will be experiencing a few of these issues as well..
Oh my goodness! Amazing article dude! Many thanks, However I am encountering issues with your RSS. I don’t understand the reason why I am unable to join it. Is there anyone else getting identical RSS issues? Anyone who knows the solution can you kindly respond? Thanx.
When I originally commented I appear to have clicked on the -Notify me when new comments are added- checkbox and now whenever a comment is added I recieve four emails with the exact same comment. Perhaps there is a way you are able to remove me from that service? Many thanks.
I used to be able to find good information from your blog posts.
After I initially commented I seem to have clicked the -Notify me when new comments are added- checkbox and from now on every time a comment is added I get 4 emails with the exact same comment. Is there a way you are able to remove me from that service? Thank you.
Thanks for the ideas shared in your blog. Something else I would like to say is that fat reduction is not about going on a celebrity diet and trying to lose as much weight as you can in a couple of weeks. The most effective way to lose weight is by getting it slowly and gradually and obeying some basic ideas which can make it easier to make the most from a attempt to shed weight. You may learn and be following most of these tips, yet reinforcing understanding never affects.
Hey there! I simply wish to offer you a big thumbs up for your great info you have got right here on this post. I am returning to your web site for more soon.
I just like the valuable information you supply in your articles. I?ll bookmark your blog and check once more here frequently. I am slightly sure I?ll be told lots of new stuff proper here! Good luck for the next!
There’s definately a lot to know about this topic. I love all the points you made.
Everything is very open with a precise description of the issues. It was really informative. Your website is very helpful. Many thanks for sharing.
Hey there! Do you know if they make any plugins to assist with Search Engine Optimization? I’m trying to get my blog to rank for some targeted keywords but I’m not seeing very good gains. If you know of any please share. Many thanks!
Undeniably imagine that which you stated. Your favorite reason appeared to be on the net the simplest thing to keep in mind of. I say to you, I certainly get annoyed at the same time as other folks consider concerns that they just do not know about. You managed to hit the nail upon the top and also defined out the entire thing without having side effect , people could take a signal. Will probably be again to get more. Thank you
I like looking through a post that will make people think. Also, thank you for allowing for me to comment.
I like it when folks get together and share thoughts. Great blog, stick with it.
After looking into a few of the blog posts on your website, I truly like your way of writing a blog. I added it to my bookmark site list and will be checking back in the near future. Take a look at my web site too and let me know how you feel.
I like it when people get together and share opinions. Great blog, stick with it.
There is certainly a lot to find out about this issue. I really like all of the points you made.
I needed to thank you for this great read!! I certainly loved every little bit of it. I have got you book-marked to look at new things you post…
Good day! I could have sworn I’ve been to this blog before but after looking at a few of the posts I realized it’s new to me. Anyhow, I’m definitely happy I discovered it and I’ll be book-marking it and checking back often!
Good information. Lucky me I found your blog by chance (stumbleupon). I have saved it for later!
There is definately a great deal to know about this issue. I really like all of the points you’ve made.
Good site you have got here.. It’s hard to find high-quality writing like yours nowadays. I honestly appreciate individuals like you! Take care!!
Very good info. Lucky me I recently found your website by accident (stumbleupon). I have book-marked it for later.
Your style is really unique compared to other folks I have read stuff from. I appreciate you for posting when you have the opportunity, Guess I’ll just book mark this blog.
Howdy! I could have sworn I’ve been to this site before but after browsing through many of the articles I realized it’s new to me. Nonetheless, I’m definitely delighted I found it and I’ll be bookmarking it and checking back often!
Your style is unique compared to other people I have read stuff from. Many thanks for posting when you’ve got the opportunity, Guess I will just bookmark this page.
Hi, i think that i saw you visited my web site so i came to ?return the favor?.I’m attempting to find things to enhance my site!I suppose its ok to use some of your ideas!!
Thanks for sharing your ideas. The first thing is that students have an alternative between national student loan and a private student loan where it’s easier to select student loan consolidation than over the federal student loan.
Hello there, simply turned into aware of your weblog thru Google, and located that it is truly informative. I am gonna watch out for brussels. I will be grateful for those who continue this in future. Numerous other people can be benefited from your writing. Cheers!
Appreciating the time and effort you put into your site and in depth information you provide. It’s awesome to come across a blog every once in a while that isn’t the same unwanted rehashed material. Great read! I’ve saved your site and I’m including your RSS feeds to my Google account.
May I simply say what a relief to discover someone that actually understands what they are talking about online. You definitely understand how to bring an issue to light and make it important. More and more people ought to check this out and understand this side of your story. I can’t believe you’re not more popular because you certainly possess the gift.
I like reading a post that can make people think. Also, many thanks for allowing me to comment.
Valuable info. Fortunate me I found your site by chance, and I am stunned why this twist of fate did not took place in advance! I bookmarked it.
Great site you have got here.. It’s difficult to find high-quality writing like yours nowadays. I seriously appreciate people like you! Take care!!
I would like to thnkx for the efforts you’ve put in writing this blog. I’m hoping the same high-grade blog post from you in the upcoming as well. Actually your creative writing abilities has inspired me to get my own web site now. Actually the blogging is spreading its wings quickly. Your write up is a great example of it.
I needed to thank you for this great read!! I definitely enjoyed every bit of it. I have you book marked to look at new stuff you post…
Greetings! Very helpful advice in this particular post! It is the little changes which will make the biggest changes. Thanks a lot for sharing!
After looking into a handful of the blog articles on your website, I honestly like your technique of blogging. I book-marked it to my bookmark webpage list and will be checking back soon. Please check out my website as well and tell me your opinion.
I quite like reading an article that can make people think. Also, many thanks for permitting me to comment.
This web site really has all of the information I wanted concerning this subject and didn’t know who to ask.
The next time I read a blog, Hopefully it won’t fail me just as much as this particular one. After all, I know it was my choice to read through, however I genuinely believed you would probably have something helpful to say. All I hear is a bunch of complaining about something that you can fix if you weren’t too busy seeking attention.
Can I simply say what a comfort to uncover someone who truly knows what they are talking about on the internet. You actually know how to bring an issue to light and make it important. More and more people should check this out and understand this side of the story. It’s surprising you are not more popular given that you certainly have the gift.
You are so interesting! I do not think I’ve truly read a single thing like this before. So great to discover someone with some original thoughts on this subject matter. Seriously.. thanks for starting this up. This web site is something that’s needed on the web, someone with a bit of originality.
Good post. I learn something new and challenging on sites I stumbleupon every day. It will always be exciting to read content from other writers and use something from their websites.
This website was… how do I say it? Relevant!! Finally I have found something which helped me. Thanks a lot!
There is certainly a lot to find out about this issue. I love all the points you have made.
I blog frequently and I really appreciate your information. The article has truly peaked my interest. I’m going to bookmark your site and keep checking for new information about once a week. I subscribed to your Feed too.
Thanks a ton for your post. I want to comment that the expense of car insurance will vary from one insurance policy to another, since there are so many different facets which contribute to the overall cost. For example, the make and model of the car or truck will have a tremendous bearing on the price. A reliable older family motor vehicle will have a less expensive premium when compared to a flashy racecar.
I think that is one of the most significant information for me. And i am glad studying your article. However should observation on few common issues, The site taste is great, the articles is in point of fact great : D. Good process, cheers
I like what you guys are up too. Such smart work and reporting! Keep up the superb works guys I?ve incorporated you guys to my blogroll. I think it’ll improve the value of my web site 🙂
Greetings! Very useful advice in this particular article! It is the little changes that will make the greatest changes. Thanks a lot for sharing!
Hi! I could have sworn I’ve been to this site before but after going through many of the articles I realized it’s new to me. Nonetheless, I’m certainly delighted I found it and I’ll be bookmarking it and checking back often!
excellent issues altogether, you just gained a new reader. What would you suggest in regards to your publish that you just made a few days in the past? Any positive?
Very good post. I certainly appreciate this website. Keep writing!
An outstanding share! I’ve just forwarded this onto a co-worker who had been doing a little homework on this. And he in fact bought me dinner because I found it for him… lol. So allow me to reword this…. Thanks for the meal!! But yeah, thanks for spending time to discuss this topic here on your blog.
I really like reading an article that will make men and women think. Also, thank you for permitting me to comment.
I would also like to add that in case you do not already have an insurance policy otherwise you do not take part in any group insurance, chances are you’ll well gain from seeking the assistance of a health insurance broker. Self-employed or individuals with medical conditions typically seek the help of an health insurance broker. Thanks for your writing.
Greetings! Very useful advice within this post! It is the little changes that make the largest changes. Thanks for sharing!
Can I simply just say what a comfort to uncover a person that genuinely understands what they’re discussing on the net. You definitely know how to bring a problem to light and make it important. More and more people really need to read this and understand this side of your story. I was surprised that you are not more popular given that you surely possess the gift.
After I initially left a comment I appear to have clicked the -Notify me when new comments are added- checkbox and now each time a comment is added I get 4 emails with the exact same comment. Perhaps there is a means you can remove me from that service? Thanks.
you have got a terrific weblog here! would you wish to make some invite posts on my blog?
You have made some good points there. I looked on the internet to find out more about the issue and found most people will go along with your views on this website.
Hello there! This article could not be written any better! Going through this article reminds me of my previous roommate! He continually kept preaching about this. I am going to send this information to him. Pretty sure he will have a good read. I appreciate you for sharing!
Good post. I learn something totally new and challenging on blogs I stumbleupon every day. It’s always helpful to read content from other authors and use something from their sites.
I like reading a post that can make men and women think. Also, many thanks for allowing me to comment.
This is a topic that is near to my heart… Many thanks! Where can I find the contact details for questions?
Wonderful article! We will be linking to this great content on our site. Keep up the good writing.
May I simply just say what a comfort to discover somebody who genuinely understands what they’re talking about on the internet. You definitely know how to bring an issue to light and make it important. More and more people have to read this and understand this side of your story. I was surprised that you aren’t more popular since you certainly possess the gift.
Hi! I could have sworn I’ve been to this website before but after going through a few of the articles I realized it’s new to me. Anyhow, I’m certainly happy I stumbled upon it and I’ll be bookmarking it and checking back often!
Oh my goodness! Awesome article dude! Thank you, However I am experiencing difficulties with your RSS. I don’t know the reason why I cannot subscribe to it. Is there anybody getting identical RSS issues? Anybody who knows the solution will you kindly respond? Thanx!!
Thanks for your post. My spouse and i have constantly observed that the majority of people are wanting to lose weight because they wish to look slim in addition to looking attractive. Nonetheless, they do not continually realize that there are many benefits for losing weight additionally. Doctors say that over weight people come across a variety of disorders that can be perfectely attributed to their excess weight. The good thing is that people who definitely are overweight along with suffering from different diseases are able to reduce the severity of the illnesses by means of losing weight. You possibly can see a steady but identifiable improvement with health as soon as even a negligible amount of fat loss is obtained.
I have seen plenty of useful things on your internet site about personal computers. However, I’ve got the viewpoint that lap tops are still not quite powerful adequately to be a wise decision if you usually do projects that require loads of power, such as video enhancing. But for net surfing, statement processing, and the majority of other popular computer functions they are fine, provided you do not mind the screen size. Many thanks for sharing your opinions.
I’ve noticed that credit restoration activity must be conducted with tactics. If not, it’s possible you’ll find yourself causing harm to your position. In order to succeed in fixing to your credit rating you have to be careful that from this second you pay all your monthly dues promptly prior to their appointed date. It really is significant for the reason that by not accomplishing this, all other methods that you will decide to use to improve your credit position will not be successful. Thanks for expressing your suggestions.
Can I just say what a relief to uncover someone that genuinely knows what they’re talking about over the internet. You definitely realize how to bring an issue to light and make it important. A lot more people need to look at this and understand this side of your story. It’s surprising you’re not more popular since you surely have the gift.
Greetings! Very helpful advice within this post! It is the little changes which will make the most important changes. Thanks for sharing!
Oh my goodness! Awesome article dude! Thank you, However I am going through difficulties with your RSS. I don’t understand why I am unable to subscribe to it. Is there anybody else having identical RSS issues? Anyone that knows the answer can you kindly respond? Thanx.
Your style is so unique in comparison to other folks I have read stuff from. Thank you for posting when you’ve got the opportunity, Guess I will just bookmark this page.
You’ve made some really good points there. I checked on the internet for additional information about the issue and found most individuals will go along with your views on this web site.
Excellent post! We are linking to this particularly great content on our website. Keep up the good writing.
Good day! I could have sworn I’ve been to this web site before but after browsing through some of the articles I realized it’s new to me. Anyways, I’m definitely pleased I found it and I’ll be bookmarking it and checking back regularly!
I seriously love your website.. Pleasant colors & theme. Did you make this website yourself? Please reply back as I’m hoping to create my own personal blog and would love to find out where you got this from or just what the theme is named. Cheers!
This is a topic that’s close to my heart… Take care! Exactly where are your contact details though?
Pretty! This has been a really wonderful article. Thanks for providing this information.
Thanks for your posting. I have always noticed that many people are eager to lose weight simply because wish to look slim as well as attractive. Nevertheless, they do not generally realize that there are many benefits for losing weight as well. Doctors claim that fat people come across a variety of diseases that can be perfectely attributed to their own excess weight. The great thing is that people who’re overweight and also suffering from different diseases are able to reduce the severity of their particular illnesses by means of losing weight. It’s possible to see a steady but noticeable improvement with health when even a bit of a amount of weight loss is attained.
Everything is very open with a really clear description of the challenges. It was really informative. Your website is extremely helpful. Thank you for sharing.
I’m amazed, I must say. Rarely do I encounter a blog that’s both educative and engaging, and let me tell you, you have hit the nail on the head. The issue is an issue that not enough men and women are speaking intelligently about. Now i’m very happy that I stumbled across this in my hunt for something concerning this.
I have learned some significant things through your blog post. One other point I would like to state is that there are numerous games available on the market designed in particular for toddler age children. They include things like pattern identification, colors, pets, and shapes. These usually focus on familiarization as an alternative to memorization. This makes children and kids engaged without having a sensation like they are learning. Thanks
Excellent web site you have here.. It’s hard to find high quality writing like yours these days. I honestly appreciate people like you! Take care!!
Thanks a lot for the helpful write-up. It is also my opinion that mesothelioma cancer has an particularly long latency phase, which means that symptoms of the disease may well not emerge until finally 30 to 50 years after the first exposure to mesothelioma. Pleural mesothelioma, that’s the most common sort and affects the area within the lungs, will cause shortness of breath, upper body pains, as well as a persistent coughing, which may cause coughing up blood vessels.
Pretty! This has been an extremely wonderful article. Thank you for supplying this information.
There’s certainly a great deal to find out about this topic. I love all of the points you’ve made.
I used to be able to find good advice from your articles.
This is a topic that’s near to my heart… Many thanks! Where can I find the contact details for questions?
I used to be able to find good advice from your blog articles.
Good information. Lucky me I found your website by chance (stumbleupon). I have saved as a favorite for later.
Hello There. I discovered your weblog using msn. This is a really neatly written article. I?ll be sure to bookmark it and return to read extra of your useful information. Thank you for the post. I will certainly return.
I should say also believe that mesothelioma cancer is a rare form of many forms of cancer that is normally found in individuals previously familiar with asbestos. Cancerous tissues form while in the mesothelium, which is a protective lining which covers a lot of the body’s body organs. These cells typically form from the lining of your lungs, mid-section, or the sac which encircles one’s heart. Thanks for expressing your ideas.
Thanks for sharing your ideas. I’d also like to say that video games have been ever evolving. Better technology and revolutions have assisted create genuine and fun games. Most of these entertainment video games were not that sensible when the concept was first being attempted. Just like other designs of electronics, video games as well have had to progress by many years. This is testimony towards the fast progression of video games.
Oh my goodness! an incredible article dude. Thank you However I am experiencing concern with ur rss . Don?t know why Unable to subscribe to it. Is there anyone getting identical rss downside? Anybody who is aware of kindly respond. Thnkx
There may be noticeably a bundle to find out about this. I assume you made sure nice factors in options also.
An additional issue is that video gaming became one of the all-time most significant forms of entertainment for people of various age groups. Kids engage in video games, and adults do, too. Your XBox 360 has become the favorite video games systems for folks who love to have a huge variety of activities available to them, and also who like to learn live with some others all over the world. Many thanks for sharing your ideas.
Hi there, I believe your website may be having web browser compatibility problems. Whenever I look at your website in Safari, it looks fine but when opening in IE, it has some overlapping issues. I merely wanted to give you a quick heads up! Besides that, fantastic site!
Having read this I thought it was rather informative. I appreciate you finding the time and energy to put this article together. I once again find myself personally spending a lot of time both reading and leaving comments. But so what, it was still worth it.
You’re so cool! I do not think I’ve truly read anything like that before. So good to find someone with a few unique thoughts on this subject matter. Seriously.. thank you for starting this up. This website is one thing that is needed on the web, someone with some originality.
Thanks for your tips on this blog. One particular thing I would choose to say is purchasing electronic products items in the Internet is nothing new. In fact, in the past ten years alone, the marketplace for online consumer electronics has grown considerably. Today, you will find practically just about any electronic device and product on the Internet, ranging from cameras along with camcorders to computer elements and video gaming consoles.
This is a very good tip particularly to those fresh to the blogosphere. Short but very accurate information… Thank you for sharing this one. A must read article!
In these days of austerity in addition to relative stress about having debt, many individuals balk contrary to the idea of using a credit card in order to make acquisition of merchandise or maybe pay for any gift giving occasion, preferring, instead just to rely on the particular tried along with trusted technique of making payment – hard cash. However, if you have the cash there to make the purchase 100 , then, paradoxically, this is the best time to use the credit card for several motives.
Good post. I learn something totally new and challenging on blogs I stumbleupon every day. It’s always exciting to read content from other authors and practice a little something from their web sites.
Someone necessarily lend a hand to make critically posts I’d state. This is the first time I frequented your web page and to this point? I amazed with the research you made to create this particular put up amazing. Great task!
I’m truly enjoying the design and layout of your site. It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often. Did you hire out a developer to create your theme? Superb work!
Spot on with this write-up, I really believe that this site needs a lot more attention. I’ll probably be back again to see more, thanks for the advice.
You should be a part of a contest for one of the highest quality websites on the web. I’m going to recommend this site!
Good day! I just want to offer you a big thumbs up for your excellent information you’ve got right here on this post. I’ll be returning to your site for more soon.
Saved as a favorite, I like your web site!
hello!,I like your writing so much! share we communicate more about your article on AOL? I need a specialist on this area to solve my problem. Maybe that’s you! Looking forward to see you.
A fascinating discussion is worth comment. I do think that you need to publish more on this issue, it might not be a taboo subject but typically people don’t talk about such topics. To the next! All the best!
Another issue is that video games are normally serious anyway with the main focus on learning rather than enjoyment. Although, it comes with an entertainment factor to keep children engaged, every game is usually designed to focus on a specific group of skills or programs, such as mathmatical or scientific discipline. Thanks for your write-up.
Interesting blog post. What I would like to contribute is that personal computer memory has to be purchased when your computer can no longer cope with what you do with it. One can mount two RAM boards with 1GB each, as an illustration, but not one of 1GB and one having 2GB. One should check the car maker’s documentation for one’s PC to make sure what type of memory is necessary.
After exploring a few of the blog articles on your web page, I seriously like your technique of blogging. I bookmarked it to my bookmark site list and will be checking back soon. Please check out my website too and let me know what you think.
Way cool! Some very valid points! I appreciate you penning this post and the rest of the website is also very good.
There is certainly a lot to find out about this subject. I love all the points you made.
Hi, I do think this is an excellent blog. I stumbledupon it 😉 I will revisit once again since i have book-marked it. Money and freedom is the best way to change, may you be rich and continue to guide other people.
After looking over a few of the articles on your blog, I seriously appreciate your way of writing a blog. I bookmarked it to my bookmark webpage list and will be checking back soon. Please check out my website too and let me know how you feel.
Great info. Lucky me I ran across your blog by accident (stumbleupon). I have saved it for later.
Saved as a favorite, I really like your web site!
Excellent article. I certainly love this website. Continue the good work!
Good information. Lucky me I recently found your site by chance (stumbleupon). I’ve bookmarked it for later.
Great web site you have got here.. It’s hard to find quality writing like yours nowadays. I truly appreciate individuals like you! Take care!!
Thanks for this wonderful article. One more thing to mention is that the majority of digital cameras come equipped with any zoom lens that allows more or less of that scene for being included by way of ‘zooming’ in and out. These kind of changes in {focus|focusing|concentration|target|the a**** length will be reflected inside the viewfinder and on big display screen at the back of any camera.
I really like it when people come together and share ideas. Great blog, keep it up.
I have to thank you for the efforts you have put in writing this site. I am hoping to check out the same high-grade content by you in the future as well. In fact, your creative writing abilities has encouraged me to get my very own blog now 😉
Nice post. I learn something totally new and challenging on blogs I stumbleupon on a daily basis. It will always be interesting to read articles from other writers and use a little something from their websites.
This is a topic that’s near to my heart… Many thanks! Exactly where are your contact details though?
A motivating discussion is definitely worth comment. I believe that you ought to write more about this subject, it may not be a taboo subject but typically people do not speak about such topics. To the next! Cheers!
I must thank you for the efforts you have put in writing this site. I really hope to view the same high-grade content by you later on as well. In truth, your creative writing abilities has inspired me to get my own site now 😉
It is appropriate time to make a few plans for the longer term and it is time to be happy. I have read this publish and if I may I desire to counsel you few fascinating issues or suggestions. Maybe you can write next articles referring to this article. I want to read even more issues about it!
Hi! I could have sworn I’ve visited this blog before but after browsing through some of the articles I realized it’s new to me. Anyways, I’m definitely delighted I found it and I’ll be bookmarking it and checking back often.
Next time I read a blog, I hope that it doesn’t fail me as much as this one. After all, I know it was my choice to read, nonetheless I genuinely believed you would probably have something interesting to talk about. All I hear is a bunch of whining about something that you could fix if you were not too busy looking for attention.
This is a topic that’s near to my heart… Take care! Where are your contact details though?
bookmarked!!, I really like your blog!
Aw, this was an incredibly good post. Taking a few minutes and actual effort to generate a top notch article… but what can I say… I hesitate a lot and never seem to get anything done.
Do you mind if I quote a few of your posts as long as I provide credit and sources back to your weblog? My blog site is in the very same niche as yours and my users would really benefit from some of the information you provide here. Please let me know if this alright with you. Thank you!
Hi, I do think this is a great site. I stumbledupon it 😉 I’m going to come back once again since i have book-marked it. Money and freedom is the greatest way to change, may you be rich and continue to guide other people.
Right here is the right blog for everyone who really wants to find out about this topic. You understand a whole lot its almost tough to argue with you (not that I personally will need to…HaHa). You certainly put a brand new spin on a subject that’s been discussed for many years. Great stuff, just great.
I?ve read a few good stuff here. Certainly worth bookmarking for revisiting. I wonder how much effort you put to create such a wonderful informative site.
You are so interesting! I don’t think I’ve read anything like that before. So nice to discover another person with a few original thoughts on this subject matter. Seriously.. thank you for starting this up. This website is one thing that is needed on the web, someone with a little originality.
bookmarked!!, I like your web site!
Wow that was odd. I just wrote an really long comment but after I clicked submit my comment didn’t show up. Grrrr… well I’m not writing all that over again. Regardless, just wanted to say fantastic blog!
Very good post! We are linking to this great article on our website. Keep up the great writing.
Good information. Lucky me I came across your website by chance (stumbleupon). I’ve saved it for later.
Thank you, I’ve recently been hunting for details about this subject for ages and yours is the best I’ve found so far.
Very nice post. I just stumbled upon your blog and wished to say that I have really enjoyed browsing your blog posts. In any case I?ll be subscribing to your rss feed and I hope you write again soon!
Very good info. Lucky me I recently found your website by chance (stumbleupon). I’ve book-marked it for later.
I really like your blog.. very nice colors & theme. Did you design this website yourself or did you hire someone to do it for you? Plz answer back as I’m looking to construct my own blog and would like to know where u got this from. kudos
Your style is unique compared to other folks I have read stuff from. I appreciate you for posting when you’ve got the opportunity, Guess I will just bookmark this site.
Right here is the right web site for anybody who wants to understand this topic. You know a whole lot its almost tough to argue with you (not that I personally would want to…HaHa). You certainly put a brand new spin on a topic which has been discussed for many years. Wonderful stuff, just wonderful.
I absolutely love your site.. Very nice colors & theme. Did you create this amazing site yourself? Please reply back as I’m looking to create my very own site and would like to know where you got this from or what the theme is named. Thank you!
Through my observation, shopping for consumer electronics online may be easily expensive, nonetheless there are some guidelines that you can use to obtain the best products. There are generally ways to discover discount bargains that could help to make one to ge thet best consumer electronics products at the lowest prices. Good blog post.
I would like to thank you for the efforts you’ve put in writing this site. I am hoping the same high-grade web site post from you in the upcoming also. In fact your creative writing abilities has inspired me to get my own site now. Actually the blogging is spreading its wings fast. Your write up is a good example of it.
of course like your web-site but you have to test the spelling on quite a few of your posts. A number of them are rife with spelling issues and I to find it very bothersome to tell the truth on the other hand I?ll certainly come back again.
I have observed that online education is getting favorite because obtaining your college degree online has changed into a popular choice for many people. A large number of people have definitely not had a possibility to attend a regular college or university however seek the elevated earning potential and career advancement that a Bachelor’s Degree affords. Still others might have a diploma in one course but would choose to pursue a thing they now develop an interest in.
hi!,I love your writing so much! proportion we keep in touch more about your post on AOL? I require an expert in this house to resolve my problem. Maybe that is you! Looking ahead to see you.
Hey there! I just want to offer you a big thumbs up for your great info you have got here on this post. I’ll be coming back to your website for more soon.
I must thank you for the efforts you have put in writing this site. I really hope to see the same high-grade blog posts from you later on as well. In truth, your creative writing abilities has encouraged me to get my own blog now 😉
Greetings! Very useful advice in this particular post! It is the little changes that produce the most significant changes. Many thanks for sharing!
Greetings! Very useful advice in this particular article! It is the little changes that make the most important changes. Thanks a lot for sharing!
Your style is very unique in comparison to other people I have read stuff from. I appreciate you for posting when you have the opportunity, Guess I will just book mark this site.
What an eye-opening and thoroughly-researched article! The author’s attention to detail and ability to present complicated ideas in a understandable manner is truly admirable. I’m totally impressed by the breadth of knowledge showcased in this piece. Thank you, author, for providing your wisdom with us. This article has been a real game-changer!
It’s hard to come by experienced people on this topic, but you sound like you know what you’re talking about! Thanks
There’s definately a lot to find out about this issue. I really like all of the points you made.
Hi, I do think this is a great site. I stumbledupon it 😉 I will come back once again since I bookmarked it. Money and freedom is the greatest way to change, may you be rich and continue to guide others.
I was very pleased to discover this web site. I need to to thank you for ones time for this fantastic read!! I definitely loved every part of it and i also have you book-marked to see new stuff in your site.
Hmm is anyone else having problems with the images on this blog loading? I’m trying to find out if its a problem on my end or if it’s the blog. Any suggestions would be greatly appreciated.
Greetings! Very helpful advice within this article! It’s the little changes that will make the biggest changes. Thanks for sharing!
When I initially left a comment I seem to have clicked on the -Notify me when new comments are added- checkbox and now whenever a comment is added I get four emails with the exact same comment. There has to be a means you can remove me from that service? Many thanks.
I have seen that expenses for on-line degree specialists tend to be an incredible value. For instance a full College Degree in Communication with the University of Phoenix Online consists of Sixty credits with $515/credit or $30,900. Also American Intercontinental University Online comes with a Bachelors of Business Administration with a entire school requirement of 180 units and a price of $30,560. Online degree learning has made getting the education far more easy because you can certainly earn your own degree from the comfort of your abode and when you finish from work. Thanks for all your other tips I have learned from your web site.
When I initially left a comment I seem to have clicked on the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I get 4 emails with the same comment. There has to be a means you are able to remove me from that service? Appreciate it.
That is a really good tip especially to those fresh to the blogosphere. Simple but very precise information… Appreciate your sharing this one. A must read post.
Thanks for your posting. One other thing is always that individual American states have their own personal laws of which affect property owners, which makes it quite hard for the the nation’s lawmakers to come up with a different set of rules concerning property foreclosures on homeowners. The problem is that every state features own laws and regulations which may have interaction in an adverse manner when it comes to foreclosure policies.
That is a really good tip particularly to those new to the blogosphere. Short but very precise information… Thanks for sharing this one. A must read post!
Good article! We will be linking to this particularly great post on our website. Keep up the good writing.
I wanted to thank you for this good read!! I absolutely enjoyed every little bit of it. I’ve got you saved as a favorite to check out new stuff you post…
Everyone loves it when folks get together and share views. Great blog, keep it up!
Very good information. Lucky me I came across your site by accident (stumbleupon). I have book-marked it for later.
Please let me know if you’re looking for a author for your weblog. You have some really good articles and I feel I would be a good asset. If you ever want to take some of the load off, I’d really like to write some material for your blog in exchange for a link back to mine. Please blast me an e-mail if interested. Many thanks!
bookmarked!!, I love your blog.
Pretty! This has been a really wonderful article. Thank you for providing this info.
This excellent website definitely has all of the info I needed about this subject and didn’t know who to ask.
There’s certainly a great deal to find out about this topic. I like all the points you have made.
Aw, this was an exceptionally good post. Spending some time and actual effort to generate a very good article… but what can I say… I put things off a lot and never seem to get nearly anything done.
You’ve made some really good points there. I checked on the internet for additional information about the issue and found most individuals will go along with your views on this site.
Howdy, I think your web site may be having internet browser compatibility problems. Whenever I take a look at your web site in Safari, it looks fine however, if opening in Internet Explorer, it has some overlapping issues. I merely wanted to provide you with a quick heads up! Besides that, excellent website.
You’re so cool! I don’t think I have read through anything like this before. So good to discover someone with a few original thoughts on this subject. Seriously.. thank you for starting this up. This site is one thing that’s needed on the web, someone with a little originality.
bookmarked!!, I love your site!
Excellent article. I am going through a few of these issues as well..
You have made some good points there. I looked on the internet to learn more about the issue and found most individuals will go along with your views on this web site.
Hi! I simply wish to give you a big thumbs up for your great information you have got right here on this post. I am coming back to your site for more soon.
Hello there! I just want to give you a huge thumbs up for the excellent info you’ve got here on this post. I am returning to your website for more soon.
Hi! I could have sworn I’ve visited this site before but after going through many of the posts I realized it’s new to me. Regardless, I’m definitely delighted I came across it and I’ll be bookmarking it and checking back often.
Having read this I thought it was very enlightening. I appreciate you taking the time and energy to put this informative article together. I once again find myself spending a lot of time both reading and posting comments. But so what, it was still worthwhile.
I’m impressed, I must say. Seldom do I encounter a blog that’s both educative and amusing, and let me tell you, you’ve hit the nail on the head. The problem is something which not enough people are speaking intelligently about. I am very happy I found this during my search for something concerning this.
Pretty! This has been an extremely wonderful post. Thank you for supplying this information.
I really like it when individuals come together and share opinions. Great site, continue the good work.
I used to be able to find good info from your articles.
I want to to thank you for this very good read!! I definitely loved every little bit of it. I have got you saved as a favorite to look at new stuff you post…
Excellent article! We are linking to this great post on our website. Keep up the great writing.
Hello there! I simply would like to offer you a huge thumbs up for the excellent information you’ve got right here on this post. I will be returning to your site for more soon.
Someone essentially help to make seriously posts I would state. This is the first time I frequented your web page and thus far? I amazed with the research you made to create this particular publish incredible. Great job!
This site was… how do you say it? Relevant!! Finally I’ve found something that helped me. Kudos!
This is the right site for everyone who really wants to understand this topic. You realize so much its almost tough to argue with you (not that I personally would want to…HaHa). You certainly put a fresh spin on a topic that’s been discussed for ages. Wonderful stuff, just great.
Your style is very unique compared to other people I have read stuff from. Thank you for posting when you have the opportunity, Guess I’ll just book mark this page.
Saved as a favorite, I love your blog.
This blog was… how do you say it? Relevant!! Finally I have found something which helped me. Kudos!
Our family had similar issues, thanks.
I could not refrain from commenting. Well written!
It’s hard to find knowledgeable people about this subject, however, you seem like you know what you’re talking about! Thanks
Good write-up. I absolutely appreciate this website. Stick with it!
shiokambing2
I wanted to thank you for this great read!! I absolutely loved every bit of it. I have you book marked to check out new things you post…
I enjoy looking through a post that can make men and women think. Also, thank you for allowing me to comment.
shiokambing2
You are so awesome! I don’t think I have read something like this before. So nice to discover someone with unique thoughts on this subject. Seriously.. thank you for starting this up. This web site is something that is needed on the web, someone with some originality.
Very good post! We will be linking to this great article on our website. Keep up the great writing.
I seriously love your site.. Great colors & theme. Did you make this website yourself? Please reply back as I’m wanting to create my own blog and would love to learn where you got this from or exactly what the theme is called. Appreciate it!
Saved as a favorite, I really like your site.
It’s hard to come by educated people in this particular subject, however, you sound like you know what you’re talking about! Thanks
Great site you have got here.. It’s difficult to find high-quality writing like yours these days. I truly appreciate individuals like you! Take care!!
shiokambing2
bookmarked!!, I love your website!
I have seen loads of useful items on your site about pcs. However, I’ve the view that netbooks are still not quite powerful more than enough to be a option if you typically do tasks that require a lot of power, such as video enhancing. But for website surfing, statement processing, and majority of other prevalent computer functions they are okay, provided you never mind your little friend screen size. Appreciate sharing your thinking.
I have read some good stuff here. Certainly worth bookmarking for revisiting. I wonder how much effort you put to create such a wonderful informative website.
Nice post. I study something more challenging on totally different blogs everyday. It should at all times be stimulating to learn content from other writers and observe a little bit one thing from their store. I?d prefer to make use of some with the content on my blog whether or not you don?t mind. Natually I?ll offer you a link in your net blog. Thanks for sharing.
When I originally left a comment I seem to have clicked on the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I get 4 emails with the exact same comment. Is there an easy method you can remove me from that service? Kudos.
Excellent post! We will be linking to this particularly great content on our website. Keep up the great writing.
I’m very pleased to find this page. I need to to thank you for your time just for this fantastic read!! I definitely loved every little bit of it and I have you book-marked to see new stuff in your website.
Very good write-up. I certainly love this website. Thanks!
Great information. Lucky me I discovered your blog by accident (stumbleupon). I have saved it for later.
There are actually quite a lot of particulars like that to take into consideration. That could be a great point to deliver up. I supply the ideas above as normal inspiration however clearly there are questions just like the one you carry up where crucial thing might be working in trustworthy good faith. I don?t know if greatest practices have emerged round things like that, but I am certain that your job is clearly identified as a fair game. Both boys and girls feel the impression of just a moment?s pleasure, for the rest of their lives.
I’m not that much of a online reader to be honest but your blogs really nice, keep it up! I’ll go ahead and bookmark your website to come back later. Many thanks
bookmarked!!, I really like your site.
Way cool! Some very valid points! I appreciate you writing this post and also the rest of the site is extremely good.
I love it when folks get together and share opinions. Great website, keep it up!
Wonderful article! We will be linking to this great article on our website. Keep up the great writing.
The things i have often told men and women is that when looking for a good on-line electronics retail outlet, there are a few elements that you have to factor in. First and foremost, you need to make sure to choose a reputable plus reliable store that has picked up great critiques and ratings from other individuals and marketplace people. This will make sure that you are getting along with a well-known store to provide good service and support to their patrons. Thanks for sharing your thinking on this website.
I have viewed that sensible real estate agents everywhere are Marketing and advertising. They are seeing that it’s not just placing a sign post in the front place. It’s really with regards to building relationships with these dealers who someday will become purchasers. So, when you give your time and efforts to aiding these suppliers go it alone – the “Law regarding Reciprocity” kicks in. Great blog post.
Wow! This can be one particular of the most useful blogs We have ever arrive across on this subject. Basically Fantastic. I’m also a specialist in this topic therefore I can understand your hard work.
Hello! I just want to offer you a big thumbs up for your excellent information you’ve got here on this post. I’ll be returning to your blog for more soon.
Excellent article. I will be dealing with some of these issues as well..
Today, while I was at work, my sister stole my apple ipad and tested to see if it can survive a 40 foot drop, just so she can be a youtube sensation. My iPad is now destroyed and she has 83 views. I know this is totally off topic but I had to share it with someone!
Hello there! This post could not be written any better! Going through this post reminds me of my previous roommate! He always kept preaching about this. I’ll send this information to him. Fairly certain he’ll have a good read. Thanks for sharing!
I couldn’t refrain from commenting. Exceptionally well written.
dingdongtogel login
I have read a few excellent stuff here. Certainly value bookmarking for revisiting. I surprise how a lot attempt you put to make any such excellent informative web site.
You ought to take part in a contest for one of the highest quality websites on the web. I am going to highly recommend this web site!
Wonderful work! This is the type of info that should be shared around the net. Shame on the search engines for not positioning this post higher! Come on over and visit my website . Thanks =)
A motivating discussion is worth comment. I do believe that you need to write more on this subject matter, it might not be a taboo matter but generally people do not talk about such subjects. To the next! Cheers!
I’m impressed, I must say. Rarely do I come across a blog that’s both equally educative and amusing, and let me tell you, you’ve hit the nail on the head. The problem is something which too few folks are speaking intelligently about. I am very happy I found this in my search for something relating to this.
I would like to thank you for the efforts you have put in penning this blog. I really hope to check out the same high-grade blog posts by you later on as well. In truth, your creative writing abilities has encouraged me to get my own website now 😉
Everything is very open with a precise description of the issues. It was definitely informative. Your site is very useful. Many thanks for sharing.
I blog often and I really appreciate your information. This article has truly peaked my interest. I will take a note of your site and keep checking for new details about once a week. I opted in for your Feed too.
Saved as a favorite, I really like your web site!
Can I just say what a comfort to discover someone who actually knows what they are discussing on the net. You definitely know how to bring a problem to light and make it important. More people have to read this and understand this side of the story. I was surprised you’re not more popular because you most certainly have the gift.
Hello There. I found your blog the usage of msn. This is an extremely well written article. I will be sure to bookmark it and return to read extra of your useful info. Thanks for the post. I will definitely return.
Thanks for your article. What I want to point out is that when searching for a good internet electronics shop, look for a site with comprehensive information on key elements such as the security statement, safety details, any payment methods, and also other terms plus policies. Always take time to browse the help and also FAQ areas to get a far better idea of how the shop will work, what they are able to do for you, and just how you can make use of the features.
I couldn’t resist commenting. Exceptionally well written.
Pretty! This has been an extremely wonderful article. Many thanks for supplying these details.
Pretty! This was an incredibly wonderful post. Many thanks for supplying this information.
I need to to thank you for this great read!! I definitely enjoyed every bit of it. I have you bookmarked to look at new stuff you post…
Excellent read, I just passed this onto a friend who was doing some research on that. And he actually bought me lunch because I found it for him smile Therefore let me rephrase that: Thank you for lunch!
I love reading a post that can make people think. Also, many thanks for allowing me to comment.
Spot on with this write-up, I honestly think this amazing site needs a lot more attention. I’ll probably be back again to read through more, thanks for the advice!
Everything is very open with a clear description of the challenges. It was really informative. Your website is very useful. Many thanks for sharing!
Aw, this was a very good post. Spending some time and actual effort to make a great article… but what can I say… I hesitate a whole lot and never seem to get anything done.
Hello there! This blog post couldn’t be written much better! Going through this article reminds me of my previous roommate! He continually kept talking about this. I most certainly will send this post to him. Pretty sure he’ll have a good read. Thank you for sharing!
Heya i am for the first time here. I came across this board and I find It truly useful & it helped me out a lot. I hope to give something back and help others like you aided me.
You’re so interesting! I do not suppose I’ve truly read through something like that before. So wonderful to discover another person with some unique thoughts on this issue. Really.. many thanks for starting this up. This site is something that is required on the web, someone with a little originality.
This blog was… how do you say it? Relevant!! Finally I’ve found something which helped me. Many thanks!
Howdy! This blog post couldn’t be written any better! Looking at this article reminds me of my previous roommate! He constantly kept talking about this. I will forward this information to him. Pretty sure he’s going to have a very good read. I appreciate you for sharing!
I like reading an article that will make men and women think. Also, thank you for allowing me to comment.
Very nice article. I definitely appreciate this site. Stick with it!
Way cool! Some extremely valid points! I appreciate you writing this article and the rest of the site is also very good.
Thanks for the several tips contributed on this web site. I have noticed that many insurance firms offer customers generous deals if they decide to insure more and more cars with them. A significant volume of households have several cars these days, particularly those with old teenage children still located at home, plus the savings in policies might soon mount up. So it pays off to look for a bargain.
I have not checked in here for a while since I thought it was getting boring, but the last several posts are great quality so I guess I will add you back to my everyday bloglist. You deserve it my friend 🙂
Very good article. I will be facing many of these issues as well..
Pretty! This has been a really wonderful post. Thank you for providing these details.
Good day! I could have sworn I’ve been to this web site before but after looking at some of the articles I realized it’s new to me. Regardless, I’m certainly pleased I found it and I’ll be bookmarking it and checking back often.
Nice post. I learn something totally new and challenging on websites I stumbleupon everyday. It will always be helpful to read through content from other authors and practice something from their websites.
I must thank you for the efforts you’ve put in penning this site. I am hoping to view the same high-grade blog posts from you in the future as well. In truth, your creative writing abilities has motivated me to get my own, personal site now 😉
Having read this I thought it was rather enlightening. I appreciate you spending some time and energy to put this informative article together. I once again find myself personally spending a significant amount of time both reading and posting comments. But so what, it was still worth it.
Right here is the perfect blog for everyone who hopes to find out about this topic. You know so much its almost hard to argue with you (not that I really will need to…HaHa). You certainly put a brand new spin on a subject that has been discussed for a long time. Excellent stuff, just wonderful.
I’m very pleased to uncover this great site. I wanted to thank you for your time for this wonderful read!! I definitely appreciated every little bit of it and i also have you bookmarked to see new information on your website.
I do accept as true with all of the concepts you’ve presented on your post. They are really convincing and will definitely work. Still, the posts are very short for beginners. Could you please extend them a bit from next time? Thanks for the post.
I’m not that much of a internet reader to be honest but your sites really nice, keep it up! I’ll go ahead and bookmark your site to come back later on. All the best
After I originally commented I appear to have clicked the -Notify me when new comments are added- checkbox and now whenever a comment is added I receive four emails with the exact same comment. Perhaps there is a means you can remove me from that service? Thanks.
It’s perfect time to make a few plans for the longer term and it is time to be happy. I have learn this put up and if I could I wish to counsel you some fascinating issues or advice. Maybe you could write subsequent articles referring to this article. I desire to learn even more things approximately it!
Woah! I’m really enjoying the template/theme of this site. It’s simple, yet effective. A lot of times it’s tough to get that “perfect balance” between usability and visual appeal. I must say that you’ve done a very good job with this. In addition, the blog loads very quick for me on Safari. Superb Blog!
I feel that is among the so much significant info for me. And i am satisfied studying your article. However should commentary on some basic issues, The site style is ideal, the articles is in reality excellent : D. Excellent activity, cheers
This article is a breath of fresh air! The author’s unique perspective and insightful analysis have made this a truly engrossing read. I’m appreciative for the effort he has put into creating such an informative and provocative piece. Thank you, author, for providing your wisdom and sparking meaningful discussions through your brilliant writing!
Thanks for the new things you have revealed in your article. One thing I’d really like to comment on is that FSBO interactions are built as time passes. By launching yourself to the owners the first weekend their FSBO is announced, prior to masses start out calling on Thursday, you develop a good association. By mailing them resources, educational resources, free reviews, and forms, you become a great ally. Through a personal affinity for them in addition to their situation, you develop a solid connection that, in many cases, pays off in the event the owners opt with an adviser they know as well as trust — preferably you actually.
Everything is very open with a very clear description of the challenges. It was definitely informative. Your site is very useful. Thank you for sharing!
I’m amazed, I have to admit. Seldom do I encounter a blog that’s both equally educative and amusing, and without a doubt, you’ve hit the nail on the head. The issue is something that not enough people are speaking intelligently about. I’m very happy that I stumbled across this during my search for something relating to this.
After I originally commented I seem to have clicked on the -Notify me when new comments are added- checkbox and from now on each time a comment is added I recieve four emails with the exact same comment. Is there a means you are able to remove me from that service? Kudos.
This is a really good tip especially to those fresh to the blogosphere. Simple but very accurate info… Appreciate your sharing this one. A must read article.
One other issue is when you are in a situation where you don’t have a cosigner then you may really want to try to make use of all of your financing options. You will discover many awards and other scholarships or grants that will present you with finances to help you with college expenses. Many thanks for the post.
Greetings! Very useful advice in this particular post! It’s the little changes that make the most important changes. Thanks a lot for sharing!
I want to to thank you for this excellent read!! I certainly enjoyed every bit of it. I’ve got you book marked to look at new things you post…
Your style is unique compared to other folks I have read stuff from. Thanks for posting when you’ve got the opportunity, Guess I’ll just bookmark this web site.
Hi! I could have sworn I’ve been to this site before but after looking at a few of the articles I realized it’s new to me. Regardless, I’m definitely pleased I stumbled upon it and I’ll be book-marking it and checking back frequently.
After checking out a few of the blog articles on your site, I truly like your way of writing a blog. I book-marked it to my bookmark website list and will be checking back soon. Take a look at my website too and tell me what you think.
I enjoy reading an article that can make men and women think. Also, thanks for permitting me to comment.
An additional issue is that video games are usually serious anyway with the main focus on knowing things rather than fun. Although, it comes with an entertainment facet to keep your children engaged, each game will likely be designed to work towards a specific experience or programs, such as mathmatical or research. Thanks for your article.
You’re so cool! I do not think I’ve truly read through something like that before. So great to find someone with original thoughts on this issue. Seriously.. thanks for starting this up. This web site is something that’s needed on the web, someone with a little originality.
Hey! This is my first comment here so I just wanted to give a quick shout out and tell you I really enjoy reading your articles. Can you recommend any other blogs/websites/forums that cover the same subjects? Appreciate it!
Can I just say what a relief to uncover someone that really understands what they’re discussing on the net. You certainly realize how to bring an issue to light and make it important. A lot more people really need to read this and understand this side of the story. I can’t believe you are not more popular given that you definitely have the gift.
Everyone loves it when individuals get together and share opinions. Great blog, keep it up.
I was able to find good advice from your blog posts.
Very good article. I’m going through a few of these issues as well..
Right here is the perfect web site for anyone who really wants to find out about this topic. You know so much its almost hard to argue with you (not that I actually will need to…HaHa). You certainly put a fresh spin on a subject which has been discussed for a long time. Wonderful stuff, just wonderful.
That is a really good tip particularly to those new to the blogosphere. Brief but very accurate info… Thanks for sharing this one. A must read article!
affordablecanvaspaintings.com.au is Australia Popular Online 100 percent Handmade Art Store. We deliver Budget Handmade Canvas Paintings, Abstract Art, Oil Paintings, Artwork Sale, Acrylic Wall Art Paintings, Custom Art, Oil Portraits, Pet Paintings, Building Paintings etc. 1000+ Designs To Choose From, Highly Experienced Artists team, Up-to 50 percent OFF SALE and FREE Delivery Australia, Sydney, Melbourne, Brisbane, Adelaide, Hobart and all regional areas. We ship worldwide international locations. Order Online Your Handmade Art Today.
Hiya, I am really glad I have found this information. Nowadays bloggers publish only about gossip and net stuff and this is actually frustrating.
affordablecanvaspaintings.com.au is Australia Popular Online 100 percent Handmade Art Store. We deliver Budget Handmade Canvas Paintings, Abstract Art, Oil Paintings, Artwork Sale, Acrylic Wall Art Paintings, Custom Art, Oil Portraits, Pet Paintings, Building Paintings etc. 1000+ Designs To Choose From, Highly Experienced Artists team, Up-to 50 percent OFF SALE and FREE Delivery Australia, Sydney, Melbourne, Brisbane, Adelaide, Hobart and all regional areas. We ship worldwide international locations. Order Online Your Handmade Art Today.
Thank you a bunch for sharing this with all people you really know what you are speaking about! Bookmarked. Please additionally talk over with my website =). We may have a link alternate arrangement among us!
deniz kucukkay
dr basat
terziler
ahd clinic
adem köse
basat
terziler
ahd clinic
Spot on with this write-up, I honestly believe that this amazing site needs much more attention. I’ll probably be returning to read through more, thanks for the information.
Aw, this was a very good post. Spending some time and actual effort to make a top notch article… but what can I say… I put things off a whole lot and don’t manage to get nearly anything done.
Hi there, I believe your site might be having internet browser compatibility issues. When I look at your web site in Safari, it looks fine however, when opening in Internet Explorer, it’s got some overlapping issues. I just wanted to provide you with a quick heads up! Aside from that, fantastic blog!
Aw, this was an incredibly nice post. Finding the time and actual effort to create a superb article… but what can I say… I hesitate a whole lot and don’t seem to get nearly anything done.
Once I originally commented I clicked the -Notify me when new feedback are added- checkbox and now each time a remark is added I get 4 emails with the same comment. Is there any approach you may remove me from that service? Thanks!
Excellent post. I will be facing a few of these issues as well..
This site definitely has all of the information I needed concerning this subject and didn’t know who to ask.
You are so cool! I don’t think I’ve truly read something like that before. So wonderful to discover another person with unique thoughts on this subject. Seriously.. thank you for starting this up. This site is something that is needed on the internet, someone with a bit of originality.
Can I simply just say what a relief to find someone that truly understands what they are discussing on the internet. You certainly realize how to bring a problem to light and make it important. More and more people have to read this and understand this side of your story. I was surprised that you are not more popular given that you certainly possess the gift.
I was very happy to find this great site. I wanted to thank you for ones time for this fantastic read!! I definitely savored every little bit of it and I have you saved as a favorite to see new stuff on your blog.
After I originally left a comment I seem to have clicked on the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I recieve four emails with the same comment. Perhaps there is a means you can remove me from that service? Cheers.
May I just say what a comfort to discover someone that really knows what they’re talking about over the internet. You actually know how to bring an issue to light and make it important. More people ought to check this out and understand this side of your story. I was surprised you are not more popular because you most certainly possess the gift.
doktor kbb
This is a great blog. Thank you for the very informative post.
After I originally commented I seem to have clicked the -Notify me when new comments are added- checkbox and now every time a comment is added I get 4 emails with the exact same comment. Perhaps there is a means you can remove me from that service? Thank you.
Hi, I do believe this is a great web site. I stumbledupon it 😉 I’m going to revisit yet again since i have book-marked it. Money and freedom is the best way to change, may you be rich and continue to help other people.
Greetings! Very useful advice within this article! It is the little changes that make the most important changes. Thanks a lot for sharing!
Spot on with this write-up, I seriously believe this amazing site needs much more attention. I’ll probably be returning to read through more, thanks for the information.
Nice post. I learn something new and challenging on websites I stumbleupon every day. It will always be interesting to read through articles from other writers and use something from other web sites.
This is a topic which is close to my heart… Thank you! Exactly where can I find the contact details for questions?
Everything is very open with a precise clarification of the issues. It was definitely informative. Your website is very useful. Thanks for sharing!
Your style is very unique in comparison to other folks I’ve read stuff from. I appreciate you for posting when you’ve got the opportunity, Guess I will just book mark this site.
Good article! We are linking to this particularly great content on our site. Keep up the great writing.
In the grand scheme of things you actually secure a B+ with regard to effort and hard work. Where exactly you misplaced me personally ended up being on all the particulars. As it is said, details make or break the argument.. And it couldn’t be much more correct here. Having said that, permit me say to you just what did work. The text is quite powerful and that is most likely the reason why I am taking the effort in order to comment. I do not really make it a regular habit of doing that. Second, even though I can easily notice a leaps in reason you come up with, I am not really confident of just how you seem to connect the ideas which inturn produce your final result. For now I will, no doubt subscribe to your position however hope in the foreseeable future you actually link the dots much better.
I could not refrain from commenting. Perfectly written!
I have noticed that over the course of building a relationship with real estate managers, you’ll be able to get them to understand that, in every real estate contract, a commission rate is paid. In the long run, FSBO sellers tend not to “save” the commission payment. Rather, they struggle to win the commission simply by doing the agent’s job. In accomplishing this, they spend their money plus time to execute, as best they will, the obligations of an representative. Those tasks include uncovering the home by marketing, offering the home to prospective buyers, creating a sense of buyer desperation in order to make prompt an offer, organizing home inspections, taking on qualification checks with the loan provider, supervising fixes, and facilitating the closing.
Took me time to read all the comments, but I really enjoyed the article. It proved to be Very helpful to me and I am sure to all the commenters here It’s always nice when you can not only be informed, but also entertained I’m sure you had fun writing this article.
guncel ozturk
This is the precise weblog for anybody who wants to search out out about this topic. You realize a lot its virtually onerous to argue with you (not that I truly would need?HaHa). You definitely put a new spin on a topic thats been written about for years. Nice stuff, just great!
Wug8AhTF82v
I was very pleased to discover this great site. I wanted to thank you for ones time for this fantastic read!! I definitely liked every little bit of it and i also have you saved to fav to see new information on your site.
I would also like to add that if you do not already have got an insurance policy or maybe you do not remain in any group insurance, you will well take advantage of seeking the assistance of a health insurance broker. Self-employed or people having medical conditions typically seek the help of any health insurance dealer. Thanks for your writing.
guncel ozturk
Hi, Neat post. There’s a problem with your site in internet explorer, would test this? IE still is the market leader and a big portion of people will miss your magnificent writing because of this problem.
You are so cool! I don’t suppose I’ve truly read through something like this before. So nice to discover someone with unique thoughts on this subject matter. Really.. thanks for starting this up. This web site is one thing that is required on the web, someone with a bit of originality.
Next time I read a blog, Hopefully it won’t fail me just as much as this one. After all, I know it was my choice to read, but I genuinely thought you’d have something helpful to say. All I hear is a bunch of complaining about something that you could possibly fix if you weren’t too busy looking for attention.
Excellent post. I will be facing some of these issues as well..
This website was… how do I say it? Relevant!! Finally I’ve found something which helped me. Kudos!
Hello there, I do believe your web site could possibly be having web browser compatibility issues. When I look at your site in Safari, it looks fine however when opening in I.E., it has some overlapping issues. I merely wanted to give you a quick heads up! Aside from that, wonderful site!
After going over a number of the articles on your web page, I honestly appreciate your way of writing a blog. I saved as a favorite it to my bookmark site list and will be checking back soon. Take a look at my website too and tell me what you think.
I love reading through an article that will make men and women think. Also, thanks for allowing for me to comment.
When I originally left a comment I seem to have clicked the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I receive 4 emails with the exact same comment. Is there a means you are able to remove me from that service? Kudos.
Pretty nice post. I just stumbled upon your blog and wanted to say that I’ve really enjoyed surfing around your blog posts. After all I will be subscribing to your rss feed and I hope you write again very soon!
Howdy! This article could not be written any better! Reading through this article reminds me of my previous roommate! He always kept talking about this. I am going to send this information to him. Pretty sure he’ll have a good read. Many thanks for sharing!
This is definitely a wonderful webpage, thanks a lot..
Hi there! I just wanted to ask if you ever have any trouble with hackers? My last blog (wordpress) was hacked and I ended up losing several weeks of hard work due to no back up. Do you have any solutions to protect against hackers?
If wings are your thing, Tinker Bell’s sexy Halloween costume design is all grown up.
Wonderful blog! I found it while searching on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Appreciate it
I like what you guys are up also. Such smart work and reporting! Keep up the superb works guys I?ve incorporated you guys to my blogroll. I think it’ll improve the value of my web site 🙂
psihoterapie online Pascani
Thanks alot : ) for your post. I’d really like to comment that the tariff of car insurance differs a lot from one plan to another, for the reason that there are so many different issues which bring about the overall cost. One example is, the brand name of the automobile will have a significant bearing on the fee. A reliable aged family motor vehicle will have a less expensive premium compared to a flashy sports car.
Thank you for this article. I will also like to express that it can become hard when you are in school and starting out to initiate a long history of credit. There are many students who are merely trying to endure and have a long or favourable credit history can sometimes be a difficult point to have.
Hi there! Quick question that’s entirely off topic. Do you know how to make your site mobile friendly? My web site looks weird when viewing from my iphone 4. I’m trying to find a theme or plugin that might be able to correct this issue. If you have any suggestions, please share. Thanks!
Thanks for your beneficial post. Through the years, I have come to understand that the symptoms of mesothelioma are caused by the particular build up associated fluid regarding the lining of the lung and the breasts cavity. The illness may start within the chest location and propagate to other areas of the body. Other symptoms of pleural mesothelioma cancer include fat reduction, severe respiration trouble, fever, difficulty ingesting, and infection of the neck and face areas. It ought to be noted that some people existing with the disease tend not to experience any kind of serious signs and symptoms at all.
certainly like your web site however you need to check the spelling on quite a few of your posts. Many of them are rife with spelling problems and I to find it very troublesome to inform the truth however I will definitely come again again.
There is so much to try to understand
Hi, I think your blog might be having browser compatibility issues. When I look at your website in Opera, it looks fine but when opening in Internet Explorer, it has some overlapping. I just wanted to give you a quick heads up! Other then that, amazing blog!
I’ve learned some important things via your post. I’d personally also like to state that there will be a situation that you will make application for a loan and do not need a co-signer such as a Federal Student Aid Loan. But when you are getting financing through a standard loan company then you need to be willing to have a co-signer ready to help you. The lenders will certainly base their decision on the few variables but the most significant will be your credit standing. There are some lenders that will furthermore look at your work history and make a decision based on this but in many cases it will hinge on your credit score.
I’m usually to blogging and i actually recognize your content. The article has really peaks my interest. I am going to bookmark your site and keep checking for brand new information.
I have learned new things through your website. One other thing I want to say is newer laptop operating systems have a tendency to allow a lot more memory to use, but they also demand more ram simply to operate. If your computer could not handle additional memory plus the newest software package requires that storage increase, it may be the time to shop for a new Computer system. Thanks
Usually I do not read article on blogs, but I would like to say that this write-up very forced me to try and do it! Your writing style has been amazed me. Thanks, very nice article.
Great blog! Do you have any tips and hints for aspiring writers? I’m hoping to start my own blog soon but I’m a little lost on everything. Would you advise starting with a free platform like WordPress or go for a paid option? There are so many choices out there that I’m totally overwhelmed .. Any ideas? Many thanks!
There’s definately a great deal to learn about this issue. I really like all the points you made.
Great items from you, man. I have keep in mind your stuff previous to and you’re simply too magnificent. I really like what you have bought right here, really like what you’re stating and the way in which through which you say it. You’re making it enjoyable and you still care for to keep it sensible. I cant wait to read much more from you. That is actually a tremendous website.
This site was… how do you say it? Relevant!! Finally I’ve found something which helped me. Appreciate it.
Would you be enthusiastic about exchanging links?
Modern and elegant design.
Thanks for this excellent article. One more thing to mention is that a lot of digital cameras arrive equipped with a new zoom lens that permits more or less of that scene to become included by simply ‘zooming’ in and out. These changes in {focus|focusing|concentration|target|the a**** length are usually reflected from the viewfinder and on significant display screen right at the back of the actual camera.
Many thanks for this article. I’d also like to convey that it can often be hard when you’re in school and just starting out to establish a long credit ranking. There are many scholars who are merely trying to endure and have a long or good credit history are often a difficult matter to have.
Thanks , I’ve recently been searching for info about this topic for ages and yours is the best I have discovered so far. But, what concerning the bottom line? Are you certain concerning the source?
I have realized that over the course of creating a relationship with real estate entrepreneurs, you’ll be able to get them to understand that, in most real estate transaction, a commission rate is paid. All things considered, FSBO sellers really don’t “save” the commission. Rather, they try to win the commission simply by doing the agent’s job. In completing this task, they expend their money in addition to time to execute, as best they could, the obligations of an realtor. Those assignments include uncovering the home via marketing, showing the home to prospective buyers, building a sense of buyer urgency in order to induce an offer, arranging home inspections, handling qualification inspections with the bank, supervising maintenance, and assisting the closing of the deal.
After I initially left a comment I appear to have clicked on the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I get four emails with the same comment. Perhaps there is an easy method you are able to remove me from that service? Cheers.
As I website possessor I believe the content matter here is rattling magnificent , appreciate it for your hard work. You should keep it up forever! Good Luck.
Thanks pertaining to discussing the following superb written content on your site. I ran into it on the search engines. I will check back again if you publish extra aricles.
You appear to know so much about this, and I see you’re a published author. Thanks
Your posts provide a clear, concise description of the issues.
The fiat on-ramp tools are wide token selection and robust security.
Thanks for your short article. I would love to say that a health insurance brokerage also works for the benefit of the particular coordinators of a group insurance cover. The health insurance broker is given a summary of benefits looked for by anyone or a group coordinator. Exactly what a broker may is seek out individuals or even coordinators which in turn best complement those desires. Then he offers his recommendations and if all parties agree, the particular broker formulates binding agreement between the 2 parties.
you’ve an excellent weblog right here! would you prefer to make some invite posts on my blog?
My brother suggested I might like this web site. He was entirely right. This post actually made my day. You can not imagine simply how much time I had spent for this info! Thanks!
One thing I’d like to say is the fact before acquiring more laptop or computer memory, have a look at the machine within which it will be installed. When the machine will be running Windows XP, for instance, the memory ceiling is 3.25GB. The installation of more than this would easily constitute just a waste. Be sure that one’s motherboard can handle your upgrade amount, as well. Interesting blog post.
Thanks for the tips you have contributed here. Furthermore, I believe there are many factors which really keep your auto insurance premium straight down. One is, to think about buying motors that are in the good list of car insurance providers. Cars which might be expensive are usually more at risk of being robbed. Aside from that insurance policies are also in accordance with the value of your vehicle, so the more pricey it is, then higher the particular premium you only pay.
I think other website proprietors should take this web site as an model, very clean and excellent user genial style and design, as well as the content. You are an expert in this topic!
One thing I want to comment on is that fat reduction plan fast can be carried out by the suitable diet and exercise. Ones size not only affects the look, but also the quality of life. Self-esteem, major depression, health risks, and also physical capabilities are afflicted in extra weight. It is possible to do everything right whilst still having a gain. Should this happen, a condition may be the culprit. While an excessive amount food and not enough work out are usually guilty, common medical ailments and widespread prescriptions might greatly increase size. Kudos for your post right here.
I have learned result-oriented things as a result of your site. One other thing I’d really like to say is always that newer computer system operating systems usually allow more memory to be utilized, but they as well demand more ram simply to run. If someone’s computer is not able to handle additional memory along with the newest computer software requires that memory increase, it can be the time to shop for a new Laptop. Thanks
I’d like to be able to write like this, but taking the time and developing articles is hard…. Takes a lot of effort.
I can’t express how much I admire the effort the author has put into creating this remarkable piece of content. The clarity of the writing, the depth of analysis, and the plethora of information provided are simply impressive. His passion for the subject is apparent, and it has definitely struck a chord with me. Thank you, author, for offering your knowledge and enlightening our lives with this exceptional article!
Good day! I could have sworn I’ve visited this web site before but after browsing through many of the posts I realized it’s new to me. Nonetheless, I’m definitely pleased I found it and I’ll be book-marking it and checking back often!
Great write-up, I?m regular visitor of one?s website, maintain up the nice operate, and It is going to be a regular visitor for a lengthy time.
Might we expect to see more of these same problems in the future?
Howdy! I could have sworn I’ve been to this blog before but after browsing through many of the posts I realized it’s new to me. Regardless, I’m certainly pleased I discovered it and I’ll be book-marking it and checking back often!
sapphire hair clinic
deniz küçükkaya
noyan
istanbul hair
mehmet emre dinç
clinicplast
medart
Good site! I truly love how it is easy on my eyes it is. I am wondering how I might be notified when a new post has been made. I’ve subscribed to your RSS which may do the trick? Have a great day!
Another issue is that video games can be serious anyway with the primary focus on mastering rather than fun. Although, there’s an entertainment feature to keep children engaged, just about every game is frequently designed to focus on a specific skill set or area, such as numbers or scientific discipline. Thanks for your write-up.
The clarity in your post is just nice and I can tell you are an expert in the subject matter.
My brother suggested I might like this web site. He was entirely right. This post actually made my day. You can not imagine simply how much time I had spent for this info! Thanks!
I really like your wp design, wherever do you down load it from?
This is one very informative blog. I like the way you write and I will bookmark your blog to my favorites.
It?s really a nice and useful piece of info. I?m glad that you shared this helpful info with us. Please keep us informed like this. Thank you for sharing.
I used to be more than happy to seek out this internet-site.I needed to thanks for your time for this excellent learn!! I definitely having fun with every little little bit of it and I’ve you bookmarked to check out new stuff you weblog post.
Hello my loved one! I wish to say that this article is awesome, great written and come with almost all important infos. I?d like to see more posts like this .
Hello.This article was extremely interesting, particularly since I was searching for thoughts on this subject last couple of days.
I can’t go into details, but I have to say its a good article!
maral
I am glad to be a visitor of this perfect blog !, appreciate it for this rare info!
If you are looking to post free classifieds in India online, we highly recommend Xpdea Classifieds. Xpdea is India’s leading free online ads posting site.
If you are looking to post free classifieds in India online, we highly recommend Xpdea Classifieds. Xpdea is India’s leading free online ads posting site.
Thanks a lot for sharing this with all people you really know what you are talking approximately! Bookmarked. Kindly also visit my website =). We will have a hyperlink exchange arrangement between us!
If you are looking to post free classifieds in India online, we highly recommend Xpdea Classifieds. Xpdea is India’s leading free online ads posting site.
If you are looking to post free classifieds in India online, we highly recommend Xpdea Classifieds. Xpdea is India’s leading free online ads posting site.
I should say also believe that mesothelioma cancer is a exceptional form of melanoma that is normally found in those previously familiar with asbestos. Cancerous tissues form while in the mesothelium, which is a protective lining that covers the majority of the body’s bodily organs. These cells normally form inside the lining in the lungs, abdominal area, or the sac which actually encircles one’s heart. Thanks for discussing your ideas.
Thanks for your posting. Another item is that being photographer will involve not only difficulty in capturing award-winning photographs but in addition hardships in acquiring the best dslr camera suited to your requirements and most especially issues in maintaining the grade of your camera. This is certainly very true and clear for those photography fans that are straight into capturing the actual nature’s eye-catching scenes – the mountains, the actual forests, the particular wild or the seas. Visiting these amazing places surely requires a dslr camera that can live up to the wild’s unpleasant area.
smile hair clinic
asli arcan global
smile hair clinic
dr terziler
My spouse and I absolutely love your blog and find a lot of your post’s to be exactly I’m looking for. Would you offer guest writers to write content available for you? I wouldn’t mind producing a post or elaborating on a number of the subjects you write in relation to here. Again, awesome weblog!
Hey! I simply would like to give a huge thumbs up for the good information you may have here on this post. I shall be coming again to your blog for extra soon.
What i discover troublesome is to find a weblog that may capture me for a minute however your blog is different. Bravo.
Surprisingly good post. I really found your primary webpage and additionally wanted to suggest that have essentially enjoyed searching your website blog posts. Whatever the case I’ll always be subscribing to your entire supply and I hope you jot down ever again soon!
I dont think I’ve read anything like this before. So good to find somebody with some original thoughts on this subject. cheers for starting this up. This blog is something that is needed on the web, someone with a little originality.
VIPWin nhà cái cá cược trực tuyến top 1 Châu Á, nơi cung cấp kho game đa dạng với tỷ lệ cược hấp dẫn, dịch vụ khách hàng tận tâm.
It is actually a nice and useful piece of info. I?m glad that you shared this useful info with us. Please stay us informed like this. Thanks for sharing.
One thing I’d really like to comment on is that fat reduction plan fast may be accomplished by the correct diet and exercise. A person’s size not simply affects appearance, but also the general quality of life. Self-esteem, depressive disorder, health risks, along with physical abilities are afflicted in putting on weight. It is possible to just make everything right but still gain. In such a circumstance, a problem may be the culprit. While an excessive amount of food rather than enough work out are usually the culprit, common medical ailments and widely used prescriptions can greatly add to size. Thanks alot : ) for your post in this article.
I quite like reading an article that will make people think. Also, thanks for allowing for me to comment.
This design is steller! You certainly know how to keep a reader entertained. Between your wit and your videos, I was almost moved to start my own blog (well, almost…HaHa!) Wonderful job. I really loved what you had to say, and more than that, how you presented it. Too cool!
You need to be a part of a contest for one of the most useful blogs on the web. I’m going to recommend this web site!
You really make it seem so easy with your presentation but I find this topic to be actually something that I think I would never understand. It seems too complicated and extremely broad for me. I am looking forward for your next post, I?ll try to get the hang of it!
The other day, while I was at work, my cousin stole my iPad and tested to see if it can survive a thirty foot drop, just so she can be a youtube sensation. My iPad is now broken and she has 83 views. I know this is entirely off topic but I had to share it with someone!
I like meeting utile info, this post has got me even more info!
Everything is very open with a precise explanation of the issues. It was really informative. Your website is very useful. Many thanks for sharing.
Thanks a lot for sharing this with all of us you actually know what you’re talking about! Bookmarked. Kindly also visit my site =). We could have a link exchange agreement between us!
Ten ranking prezentuje wyłącznie sprawdzone i legalne kasyna online Polska 2025, które oferują bezpieczną grę, ekskluzywne bonusy i uczciwe warunki. Znajdziesz tu szczegółowe analizy, aby świadomie wybrać najlepszą platformę dla siebie. Poniżej znajdziesz pełne opisy operatorów z naszego rankingu. Wstaw własne grafiki — obrazy wczytują się leniwie i zachowują naturalne proporcje (bez kadrowania).
When I originally commented I clicked the -Notify me when new comments are added- checkbox and now every time a comment is added I get four emails with the identical comment. Is there any approach you possibly can take away me from that service? Thanks!
Most of whatever you say happens to be astonishingly precise and that makes me wonder why I hadn’t looked at this in this light previously. Your article truly did switch the light on for me as far as this particular subject matter goes. Nonetheless there is actually just one issue I am not too comfy with and while I make an effort to reconcile that with the actual central idea of your issue, allow me see what all the rest of your readers have to point out.Very well done.
Howdy! Do you use Twitter? I’d like to follow you if that would be okay. I’m absolutely enjoying your blog and look forward to new posts.
Hey there this is kinda of off topic but I was wanting to know if blogs use WYSIWYG editors or if you have to manually code with HTML. I’m starting a blog soon but have no coding skills so I wanted to get advice from someone with experience. Any help would be enormously appreciated!
Today, while I was at work, my sister stole my apple ipad and tested to see if it can survive a 25 foot drop, just so she can be a youtube sensation. My iPad is now broken and she has 83 views. I know this is completely off topic but I had to share it with someone!
We’re a bunch of volunteers and starting a brand new scheme in our community. Your site provided us with useful info to work on. You’ve done a formidable activity and our entire group shall be thankful to you.
This web site certainly has all the information I needed concerning this subject and didn’t know who to ask.
Good post. I’m facing a few of these issues as well..
Pretty nice post. I just stumbled upon your weblog and wanted to say that I’ve really enjoyed surfing around your blog posts. After all I’ll be subscribing in your feed and I am hoping you write again very soon!
These days of austerity along with relative anxiousness about running into debt, a lot of people balk against the idea of using a credit card in order to make acquisition of merchandise or even pay for a vacation, preferring, instead only to rely on the tried in addition to trusted way of making transaction – cash. However, if you possess the cash there to make the purchase entirely, then, paradoxically, that’s the best time for you to use the cards for several reasons.
Having read this I thought it was really enlightening. I appreciate you taking the time and energy to put this short article together. I once again find myself personally spending way too much time both reading and commenting. But so what, it was still worth it.
I have been browsing on-line more than three hours as of late, yet I never found any fascinating article like yours. It?s lovely price enough for me. In my opinion, if all web owners and bloggers made just right content material as you probably did, the internet will be much more helpful than ever before.
Thanks a lot for sharing this with all of us you really know what you’re talking about! Bookmarked. Kindly also visit my site =). We could have a link exchange agreement between us!
I was reading through some of your content on this internet site and I believe this web site is very informative ! Continue posting .
I really love this article.
mehmet emre dinç
hair neva
I appreciate, cause I found just what I was looking for. You’ve ended my four day long hunt! God Bless you man. Have a great day. Bye -.
Magnificent beat ! I wish to apprentice while you amend your web site, how could i subscribe for a blog website? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast provided bright clear idea
Hello there, just became aware of your blog through Google, and found that it’s truly informative. I am gonna watch out for brussels. I?ll appreciate if you continue this in future. Many people will be benefited from your writing. Cheers!
buk
You write Formidable articles, keep up good work.
murad çeltik
ahd
güncel öztürk
I’m impressed, I need to say. Really rarely do I encounter a blog that’s both educational and entertaining, and let me tell you, you have hit the nail on the head.
taner
kandulu
ahd
nişanaşı
doktorkbb
crabs
ahd
yürük
I’m impressed, I need to say. Really rarely do I encounter a blog that’s both educational and entertaining, and let me tell you, you have hit the nail on the head.
I truly love your blog.. Very nice colors & theme. Did you create this site yourself? Please reply back as I’m trying to create my own personal site and would like to find out where you got this from or what the theme is named. Thanks.
Can I just say what a comfort to uncover a person that genuinely understands what they are talking about over the internet. You actually understand how to bring a problem to light and make it important. More and more people really need to read this and understand this side of your story. I was surprised that you are not more popular because you most certainly have the gift.
There is certainly a great deal to learn about this issue. I love all the points you made.
There are some serious financial ramifications here.
Hi there would you mind sharing which blog platform you’re using? I’m planning to start my own blog in the near future but I’m having a hard time choosing between BlogEngine/Wordpress/B2evolution and Drupal. The reason I ask is because your layout seems different then most blogs and I’m looking for something completely unique. P.S My apologies for getting off-topic but I had to ask!
I’m amazed, I have to admit. Seldom do I encounter a blog that’s both equally educative and interesting, and without a doubt, you have hit the nail on the head. The problem is an issue that not enough people are speaking intelligently about. I am very happy I found this in my hunt for something concerning this.
Hi! I simply would like to give you a big thumbs up for the great information you have here on this post. I’ll be returning to your site for more soon.
Hey would you mind stating which blog platform you’re working with? I’m looking to start my own blog in the near future but I’m having a tough time deciding between BlogEngine/Wordpress/B2evolution and Drupal. The reason I ask is because your layout seems different then most blogs and I’m looking for something unique. P.S My apologies for getting off-topic but I had to ask!
The very root of your writing whilst appearing reasonable originally, did not really sit very well with me after some time. Somewhere within the paragraphs you actually were able to make me a believer unfortunately just for a short while. I however have got a problem with your leaps in assumptions and one might do nicely to fill in those gaps. In the event that you actually can accomplish that, I would definitely be fascinated.
Please let us know when you plan to publish your book!
There is so much to try to understand
This is really interesting, You are an overly skilled blogger. I’ve joined your rss feed and look forward to in quest of extra of your excellent post. Also, I have shared your site in my social networks|
I have come to understand that expenses for online degree gurus tend to be an incredible value. Like a full 4-year college Degree in Communication in the University of Phoenix Online consists of 60 credits from $515/credit or $30,900. Also American Intercontinental University Online gives a Bachelors of Business Administration with a entire program element of 180 units and a tariff of $30,560. Online studying has made getting your certification far less difficult because you can certainly earn your degree through the comfort of your home and when you finish working. Thanks for other tips I have certainly learned through your website.
I think other web-site proprietors should take this website as an model, very clean and great user genial style and design, as well as the content. You are an expert in this topic!
Very nice post. I just stumbled upon your blog and wanted to say that I’ve really enjoyed browsing your blog posts. In any case I’ll be subscribing to your rss feed and I hope you write again soon!
Whats up very nice website!! Guy .. Beautiful .. Wonderful .. I’ll bookmark your blog and take the feeds also?I am satisfied to seek out numerous useful info right here within the put up, we want develop more techniques in this regard, thanks for sharing. . . . . .
You really make it seem so easy with your presentation but I find this matter to be actually something that I think I would never understand. It seems too complex and extremely broad for me. I am looking forward for your next post, I will try to get the hang of it!
Hi there, i read your blog from time to time and i own a similar one and i was just wondering if you get a lot of spam feedback? If so how do you reduce it, any plugin or anything you can suggest? I get so much lately it’s driving me crazy so any help is very much appreciated.
I like the helpful info you supply in your articles. I will bookmark your blog and test once more right here regularly. I’m fairly certain I?ll be told many new stuff proper here! Good luck for the following!
I just like the valuable info you supply in your articles. I?ll bookmark your blog and take a look at again right here frequently. I am rather certain I?ll be told plenty of new stuff proper right here! Best of luck for the next!
This is a perfect example of quality content. You can tell that plenty of thought when into this post. Keep it up.
I appreciate, cause I found exactly what I was looking for. You have ended my 4 day long hunt! God Bless you man. Have a great day. Bye
I must thank you for the efforts you’ve put in writing this website. I am hoping to see the same high-grade blog posts by you in the future as well. In truth, your creative writing abilities has motivated me to get my very own website now 😉
I like the helpful information you provide in your articles. I?ll bookmark your blog and check again here regularly. I’m quite sure I will learn plenty of new stuff right here! Best of luck for the next!
That is very interesting, You’re an excessively skilled blogger. I have joined your rss feed and stay up for seeking extra of your great post. Additionally, I’ve shared your web site in my social networks!
This is a perfect example of good content. You can tell that plenty of thought when into this post. Nice work.
Our communities really need to deal with this.
I truly appreciate this post. I have been looking everywhere for this! Thank goodness I found it on Bing. You have made my day! Thank you again
Excellent article! We will be linking to this great content on our site. Keep up the great writing.
Tips and tools you offer are so helpful to agencies in our community.
The clarity in your post is just nice and I can tell you are an expert in the subject matter.
I need to to thank you for this great read!! I certainly loved every little bit of it. I have got you book marked to look at new things you post…
Very good post. I definitely appreciate this website. Keep it up!
Oh my goodness! Amazing article dude! Thanks, However I am experiencing troubles with your RSS. I don’t understand the reason why I can’t join it. Is there anyone else getting similar RSS issues? Anyone who knows the solution will you kindly respond? Thanks!
You’re so interesting! I don’t suppose I’ve truly read something like this before. So great to discover someone with a few genuine thoughts on this topic. Seriously.. thanks for starting this up. This website is one thing that’s needed on the web, someone with some originality.
I’m glad I came across this page. I was looking for another website when I stumbled on it. Nice work.
Amazing article, cheers, I will bookmark you now.
Excellent article. I’m going through many of these issues as well..
I just sent this post to a bunch of my friends as I agree with most of what you’re saying here and the way you’ve presented it is awesome.
I have been curious about these trends, and you have really helped me. I have just told a few of my friends about this on FaceBook and they love your content just as much as I do.
This is a topic that is close to my heart… Many thanks! Exactly where can I find the contact details for questions?
Thank you for all the information was very accurate, just wondering if all this is possible.~
Great website. Lots of useful info here. I?m sending it to a few buddies ans additionally sharing in delicious. And certainly, thanks in your sweat!
It’s appropriate time to make some plans for the future and it is time to be happy. I’ve learn this submit and if I may just I want to recommend you some attention-grabbing things or advice. Perhaps you can write next articles relating to this article. I wish to read more things approximately it!
Thanks for your write-up. One other thing is that if you are advertising your property on your own, one of the problems you need to be alert to upfront is when to deal with property inspection accounts. As a FSBO vendor, the key concerning successfully moving your property plus saving money on real estate agent revenue is awareness. The more you know, the better your property sales effort are going to be. One area in which this is particularly crucial is reports.
You are good writer. Thank you.
Please let us know when you plan to publish your book!
You’re so interesting! I don’t believe I’ve truly read through anything like that before. So good to find somebody with some genuine thoughts on this subject matter. Seriously.. thank you for starting this up. This site is one thing that is required on the internet, someone with a bit of originality.
Some genuinely good blog posts on this site, thanks for contribution.
Good article. It is quite unfortunate that over the last years, the travel industry has already been able to to deal with terrorism, SARS, tsunamis, bird flu, swine flu, as well as the first ever true global economic downturn. Through it the industry has proven to be effective, resilient and dynamic, obtaining new solutions to deal with difficulty. There are usually fresh complications and the possiblility to which the marketplace must all over again adapt and react.
Very Interesting Information! Thank You For Thi Information!
Sweet blog! I found it while browsing on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Thank you
Great post. I was checking constantly this blog and I’m impressed! Very useful info specifically the last part 🙂 I care for such info much. I was looking for this certain information for a very long time. Thank you and good luck.
Hiya, I am really glad I have found this information. Nowadays bloggers publish only about gossip and net stuff and this is actually frustrating.
If you don’t mind, where do you host your weblog? I am looking for a very good web host and your webpage seams to be extremely fast and up most the time…
This is an awesome entry. Thank you very much for the supreme post provided! I was looking for this entry for a long time, but I wasn’t able to find a honest source.
With havin so much content do you ever run into any problems of plagorism or copyright violation? My blog has a lot of completely unique content I’ve either written myself or outsourced but it appears a lot of it is popping it up all over the internet without my agreement. Do you know any techniques to help reduce content from being ripped off? I’d really appreciate it.
Good website! I really love how it is simple on my eyes and the data are well written. I’m wondering how I could be notified when a new post has been made. I’ve subscribed to your RSS which must do the trick! Have a nice day!
Awesome post. It’s so good to see someone taking the time to share this information
Its like you read my mind! You seem to know a lot about this, like you wrote the book in it or something. I think that you can do with some pics to drive the message home a bit, but other than that, this is wonderful blog. A great read. I’ll certainly be back.
I appreciate your work, thanks for all the great blog posts.
This is really interesting, You’re a very skilled blogger. I have joined your feed and look forward to seeking more of your great post. Also, I’ve shared your web site in my social networks!
I just left a post similar to this one, but the material wasn’t as interesting as what’s here.
Nice post. It’s very well thought out and quite informative. Keep it up.
Might we expect to see more of these same problems in the future?
Abnormal this put up is totaly unrelated to what I was searching google for, but it surely used to be listed at the first page. I suppose your doing one thing proper if Google likes you adequate to place you at the first page of a non similar search.
Like the way you’ve outlined things. Easy to follow. Not cluttered.
Hi my family member! I want to say that this article is amazing, great written and include approximately all important infos. I’d like to see more posts like this .
With havin so much content do you ever run into any problems of plagorism or copyright violation? My site has a lot of completely unique content I’ve either written myself or outsourced but it seems a lot of it is popping it up all over the web without my agreement. Do you know any techniques to help stop content from being stolen? I’d certainly appreciate it.
I would really like to appreciate the endeavors you cash in on written this article. I’m going for the similar best product from you finding out in the foreseeable future as well. Actually your creative writing abilities has urged me to begin my very own blog now. Genuinely the blogging is distributing its wings rapidly. Your write down is often a fine illustration showing it.
I dont think Ive caught all the angles of this subject the way youve pointed them out. Youre a true star, a rock star man. Youve got so much to say and know so much about the subject that I think you should just teach a class about it
Good post. I study something more difficult on different blogs everyday. It’s going to always be stimulating to learn content material from other writers and observe a little bit one thing from their store. I’d prefer to use some with the content material on my blog whether you don’t mind. Natually I’ll give you a link in your web blog. Thanks for sharing.
Thank you for another excellent article. Where else could anybody get that type of info in such an ideal way of writing? I’ve a presentation next week, and I am on the look for such info.
You can definitely see your expertise in the work you write. The world hopes for even more passionate writers like you who aren’t afraid to say how they believe. Always go after your heart.
Saved as a favorite, I love your website!
Our local network of agencies has found your research so helpful.
Great post here. One thing I would really like to say is the fact most professional fields consider the Bachelors Degree as the entry level requirement for an online college degree. Although Associate Certifications are a great way to start, completing the Bachelors starts up many entrance doors to various employment goodies, there are numerous on-line Bachelor Course Programs available from institutions like The University of Phoenix, Intercontinental University Online and Kaplan. Another thing is that many brick and mortar institutions offer Online versions of their certifications but normally for a considerably higher cost than the providers that specialize in online diploma programs.
I would also like to convey that most of those who find themselves without health insurance usually are students, self-employed and people who are jobless. More than half from the uninsured are under the age of Thirty five. They do not think they are wanting health insurance because they’re young and also healthy. Its income is often spent on homes, food, in addition to entertainment. Some people that do represent the working class either entire or part-time are not given insurance by their jobs so they head out without due to the rising valuation on health insurance in the usa. Thanks for the tips you talk about through this website.
Good job for bringing something important to the internet!
It’s a comprehensive, yet fast read.
OK, you outline what is a big issue. But, can’t we develop more answers in the private sector?
Easily, the post is really the greatest on this laudable topic. I concur with your conclusions and will thirstily look forward to your future updates. Saying thank will not just be sufficient, for the wonderful c lucidity in your writing. I will instantly grab your rss feed to stay privy of any updates. Solid work and much success in your business enterprise!
Thanks for your publication. I would also love to opinion that the first thing you will need to conduct is find out if you really need credit score improvement. To do that you simply must get your hands on a replica of your credit profile. That should really not be difficult, because government makes it necessary that you are allowed to acquire one absolutely free copy of your real credit report every year. You just have to request the right people. You can either find out from the website for your Federal Trade Commission or maybe contact one of the leading credit agencies right away.
Thanks for the strategies you talk about through this website. In addition, many young women that become pregnant usually do not even seek to get medical care insurance because they are full of fearfulness they couldn’t qualify. Although some states right now require that insurers produce coverage irrespective of the pre-existing conditions. Prices on these types of guaranteed programs are usually larger, but when with the high cost of medical care bills it may be some sort of a safer way to go to protect your current financial future.
Nice blog here! Also your website loads up fast! What web host are you using? Can I get your affiliate link to your host? I wish my site loaded up as quickly as yours lol
Oh my goodness! a tremendous article dude. Thanks However I am experiencing difficulty with ur rss . Don?t know why Unable to subscribe to it. Is there anybody getting similar rss problem? Anyone who is aware of kindly respond. Thnkx
I just couldn’t go away your site prior to suggesting that I actually loved the standard info a person supply to your visitors? Is gonna be back often in order to check out new posts
This website was… how do you say it? Relevant!! Finally I’ve found something which helped me. Kudos.
okmark your blog and check again here frequently. I am quite certain I?ll learn many new stuff right here! Best of luck for the next!
Howdy! I simply wish to give a huge thumbs up for the great information you have here on this post. I will be coming again to your weblog for extra soon.
I really believe you will do well in the future I appreciate everything you have added to my knowledge base.
Some truly interesting info , well written and broadly user genial .
Good post. I learn something new and challenging on sites I stumbleupon on a daily basis. It will always be helpful to read through articles from other authors and use a little something from their sites.
What i discover troublesome is to find a weblog that may capture me for a minute however your blog is different. Bravo.
That is really fascinating, You’re an excessively skilled blogger. I’ve joined your rss feed and look forward to in the hunt for extra of your magnificent post. Additionally, I’ve shared your website in my social networks!
Woah this is just an insane amount of information, must of taken ages to compile so thanx so much for just sharing it with all of us. If your ever in any need of related information, just check out my own site!
I?m now not certain where you’re getting your information, however good topic. I must spend a while finding out much more or understanding more. Thanks for magnificent information I used to be on the lookout for this information for my mission.
This website truly has all of the info I needed concerning this subject and didn’t know who to ask.
Hi! I’m at work surfing around your blog from my new iphone 3gs! Just wanted to say I love reading through your blog and look forward to all your posts! Carry on the excellent work!
Hmm is anyone else encountering problems with the images on this blog loading? I’m trying to determine if its a problem on my end or if it’s the blog. Any responses would be greatly appreciated.
We can see that we need to develop policies to deal with this trend.
Thanks so much for this, keep up the good work 🙂
Just a quick note to express my appreciation. Take care
I will share you blog with my sis.
I’m really enjoying the design and layout of your blog. It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you hire out a developer to create your theme? Exceptional work!
I can’t express how much I admire the effort the author has put into creating this outstanding piece of content. The clarity of the writing, the depth of analysis, and the plethora of information offered are simply astonishing. His passion for the subject is apparent, and it has definitely resonated with me. Thank you, author, for sharing your knowledge and enlightening our lives with this exceptional article!
Thanks for the diverse tips discussed on this weblog. I have noticed that many insurance agencies offer customers generous discount rates if they choose to insure several cars with them. A significant number of households include several motor vehicles these days, specifically those with older teenage children still residing at home, as well as the savings in policies could soon increase. So it pays to look for a great deal.
Greetings from Colorado! I’m bored to tears at work so I decided to check out your site on my iphone during lunch break. I love the information you provide here and can’t wait to take a look when I get home. I’m shocked at how quick your blog loaded on my cell phone .. I’m not even using WIFI, just 3G .. Anyhow, good site!
Hmm is anyone else having problems with the images on this blog loading? I’m trying to find out if its a problem on my end or if it’s the blog. Any responses would be greatly appreciated.
The other day, while I was at work, my sister stole my apple ipad and tested to see if it can survive a 25 foot drop, just so she can be a youtube sensation. My iPad is now destroyed and she has 83 views. I know this is totally off topic but I had to share it with someone!
Hi my family member! I want to say that this article is awesome, nice written and come with almost all vital infos. I would like to peer more posts like this .|
There is definately a great deal to find out about this topic. I love all of the points you made.
Hello there! Do you know if they make any plugins to assist with Search Engine Optimization? I’m trying to get my blog to rank for some targeted keywords but I’m not seeing very good results. If you know of any please share. Appreciate it!
Thanks for your thoughts. One thing I’ve got noticed is the fact banks plus financial institutions know the spending patterns of consumers and as well understand that many people max out there their credit cards around the holidays. They properly take advantage of this real fact and commence flooding your current inbox as well as snail-mail box together with hundreds of no-interest APR card offers just after the holiday season concludes. Knowing that if you are like 98 of all American open public, you’ll soar at the one opportunity to consolidate credit card debt and shift balances towards 0 annual percentage rates credit cards.
With havin so much content do you ever run into any problems of plagorism or copyright infringement? My website has a lot of exclusive content I’ve either created myself or outsourced but it appears a lot of it is popping it up all over the internet without my permission. Do you know any ways to help stop content from being stolen? I’d genuinely appreciate it.
Amazing content on your website.
Oh my goodness! an incredible article dude. Thanks Nevertheless I’m experiencing difficulty with ur rss . Don?t know why Unable to subscribe to it. Is there anyone getting identical rss downside? Anybody who knows kindly respond. Thnkx
I am extremely impressed with your writing skills and also with the layout on your weblog. Is this a paid theme or did you modify it yourself? Anyway keep up the nice quality writing, it is rare to see a great blog like this one nowadays..
I’m not sure why but this site is loading incredibly slow for me. Is anyone else having this problem or is it a issue on my end? I’ll check back later on and see if the problem still exists.
I was wondering if you ever considered changing the layout of your site? Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better. Youve got an awful lot of text for only having 1 or two images. Maybe you could space it out better?
Wonderful post! We are linking to this great post on our site. Keep up the good writing.
Only a smiling visitor here to share the love (:, btw outstanding style and design .
I will right away grab your rss feed as I can’t find your e-mail subscription link or newsletter service. Do you have any? Please let me know in order that I could subscribe. Thanks.
This is an awesome entry. Thank you very much for the supreme post provided! I was looking for this entry for a long time, but I wasn’t able to find a honest source.
Wish I’d thought of this. Am in the field, but I procrastinate alot and haven’t written as much as I’d like. Thanks.
Hi! I’m at work surfing around your blog from my new iphone! Just wanted to say I love reading your blog and look forward to all your posts! Carry on the outstanding work!
I absolutely love your site.. Pleasant colors & theme. Did you create this amazing site yourself? Please reply back as I’m planning to create my own personal site and would love to know where you got this from or what the theme is called. Thanks!
Howdy great website! Does running a blog like this take a large amount of work? I’ve no understanding of coding but I had been hoping to start my own blog soon. Anyhow, if you have any suggestions or tips for new blog owners please share. I understand this is off subject however I just needed to ask. Appreciate it!|
I do agree with all the ideas you have presented in your post. They are very convincing and will certainly work. Still, the posts are very short for beginners. Could you please extend them a bit from next time? Thanks for the post.
Good post. I learn something new and challenging on websites I stumbleupon everyday. It will always be helpful to read through content from other writers and practice a little something from their web sites.
Furthermore, i believe that mesothelioma is a scarce form of cancer that is commonly found in people previously subjected to asbestos. Cancerous tissue form inside mesothelium, which is a shielding lining which covers most of the body’s areas. These cells generally form in the lining from the lungs, tummy, or the sac which actually encircles one’s heart. Thanks for revealing your ideas.
Fantastic beat ! I wish to apprentice while you amend your website, how can i subscribe for a blog site? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast offered bright clear idea
I?ve read a few good stuff here. Definitely worth bookmarking for revisiting. I surprise how much effort you put to make such a excellent informative web site.
Hello my friend! I want to say that this post is amazing, nice written and include approximately all significant infos. I?d like to see more posts like this.
I have realized that car insurance companies know the cars which are at risk from accidents and other risks. In addition they know what form of cars are prone to higher risk and also the higher risk they’ve got the higher the actual premium fee. Understanding the basic basics connected with car insurance will let you choose the right types of insurance policy that may take care of the needs you have in case you become involved in an accident. Thanks for sharing the ideas for your blog.
Pretty nice post. I simply stumbled upon your blog and wanted to say that I have really loved surfing around your blog posts. In any case I?ll be subscribing on your feed and I am hoping you write once more very soon!
Thanks for the suggestions you are sharing on this weblog. Another thing I would like to say is the fact getting hold of duplicates of your credit history in order to inspect accuracy of any detail will be the first action you have to perform in credit restoration. You are looking to freshen your credit report from damaging details faults that ruin your credit score.
Hmm it seems like your site ate my first comment (it was super long) so I guess I’ll just sum it up what I had written and say, I’m thoroughly enjoying your blog. I as well am an aspiring blog blogger but I’m still new to everything. Do you have any recommendations for rookie blog writers? I’d certainly appreciate it.
Greetings! Very helpful advice within this post! It is the little changes that produce the greatest changes. Many thanks for sharing!
Dude.. I am not much into reading, but somehow I got to read lots of articles on your blog. Its amazing how interesting it is for me to visit you very often. –
Useful info. Fortunate me I discovered your website by chance, and I am stunned why this twist of fate did not came about earlier! I bookmarked it.
Some genuinely choice content on this site, bookmarked .
Greetings, have tried to subscribe to this websites rss feed but I am having a bit of a problem. Can anyone kindly tell me what to do?’
Amazing article, cheers, I will bookmark you now.
You actually make it seem really easy with your presentation but I to find this matter to be really something which I believe I’d never understand. It kind of feels too complicated and very wide for me. I am taking a look forward on your subsequent post, I will try to get the dangle of it!
Please let me know if you’re looking for a writer for your blog. You have some really good articles and I feel I would be a good asset. If you ever want to take some of the load off, I’d absolutely love to write some material for your blog in exchange for a link back to mine. Please shoot me an email if interested. Thanks!
An outstanding share! I’ve just forwarded this onto a coworker who was conducting a little research on this. And he actually ordered me breakfast due to the fact that I discovered it for him… lol. So let me reword this…. Thank YOU for the meal!! But yeah, thanx for spending time to talk about this issue here on your internet site.
Enjoyed studying this, very good stuff, thanks.
My issues have been very similar, with my family. But, we made some different decisions. It’s complex.
Greetings! This is my first visit to your blog! We are a group of volunteers and starting a new project in a community in the same niche. Your blog provided us useful information to work on. You have done a wonderful job!
I?ve been exploring for a little for any high-quality articles or blog posts on this sort of space . Exploring in Yahoo I at last stumbled upon this site. Studying this info So i?m glad to express that I’ve an incredibly just right uncanny feeling I came upon just what I needed. I such a lot no doubt will make certain to do not disregard this site and give it a glance regularly.
I don?t even know how I ended up here, but I thought this post was good. I don’t know who you are but definitely you are going to a famous blogger if you are not already 😉 Cheers!
Oh my goodness! a tremendous article dude. Thanks Nonetheless I am experiencing challenge with ur rss . Don?t know why Unable to subscribe to it. Is there anybody getting identical rss drawback? Anybody who knows kindly respond. Thnkx
What i don’t understood is if truth be told how you are not really a lot more neatly-preferred than you might be now. You are so intelligent. You already know thus significantly relating to this topic, made me in my opinion imagine it from a lot of various angles. Its like men and women aren’t fascinated until it?s one thing to accomplish with Woman gaga! Your own stuffs great. All the time maintain it up!
Please let me know if you’re looking for a article author for your weblog. You have some really great articles and I believe I would be a good asset. If you ever want to take some of the load off, I’d love to write some content for your blog in exchange for a link back to mine. Please send me an e-mail if interested. Many thanks!
You ma’am have a way with words. Thank you very much!
have already been reading ur blog for a couple of days. really enjoy what you posted. btw i will be doing a report about this topic. do you happen to know any great websites or forums that I can find out more? thanks a lot.
I cannot thank you more than enough for the blogposts on your website. I know you set a lot of time and energy into these and truly hope you know how deeply I appreciate it. I hope I’ll do a similar thing person sooner or later.
Woah this is just an insane amount of information, must of taken ages to compile so thanx so much for just sharing it with all of us. If your ever in any need of related information, just check out my own site!
A thoughtful insight and ideas I will use on my blog. You’ve obviously spent some time on this. Congratulations
I am extremely inspired along with your writing abilities as neatly as with the structure in your weblog. Is that this a paid theme or did you modify it yourself? Anyway stay up the nice high quality writing, it is uncommon to see a nice blog like this one these days..
There’s certainly a lot to find out about this topic. I really like all the points you made.|
Hello there! I really enjoy reading your blog! If you keep making amazing posts like this I will come back every day to keep reading.
Substantially, the post is really the best on this laudable topic. I concur with your conclusions and will eagerly watch forward to your future updates.Just saying thanx will not just be enough, for the wonderful lucidity in your writing.
It?s really a great and useful piece of information. I?m glad that you shared this useful info with us. Please keep us up to date like this. Thanks for sharing.
I love what you’ve created here, this is definitely one of my favorite sites to visit.
I don’t normally comment on blogs.. But nice post! I just bookmarked your site
We can see that we need to develop policies to deal with this trend.
Can I just say what a relief to seek out someone who actually knows what theyre speaking about on the internet. You positively know find out how to bring a problem to mild and make it important. Extra individuals have to read this and perceive this side of the story. I cant believe youre not more in style because you positively have the gift.
Great info! Keep post great articles.
You are not right. I am assured. I can prove it. Write to me in PM, we will talk.
Thanks for the a new challenge you have discovered in your blog post. One thing I would really like to touch upon is that FSBO human relationships are built eventually. By launching yourself to the owners the first saturday their FSBO is actually announced, ahead of the masses commence calling on Wednesday, you produce a good link. By giving them instruments, educational products, free records, and forms, you become a good ally. If you take a personal fascination with them as well as their scenario, you make a solid network that, in many cases, pays off if the owners decide to go with an adviser they know as well as trust — preferably you actually.
I am glad to be a visitor of this thoroughgoing web blog ! , regards for this rare information! .
It’s time for communities to rally.
I really like your writing style, fantastic information, thank you for posting :D. “In every affair consider what precedes and what follows, and then undertake it.” by Epictetus.
Substantially, the post is really the best on this laudable topic. I concur with your conclusions and will eagerly watch forward to your future updates.Just saying thanx will not just be enough, for the wonderful lucidity in your writing.
When are you going to post again? You really entertain me!
We need to build frameworks and funding mechanisms.
Fantastic goods from you, man. I’ve understand your stuff previous to and you are just extremely excellent. I really like what you have acquired here, really like what you are saying and the way in which you say it. You make it enjoyable and you still care for to keep it wise. I can not wait to read much more from you. This is actually a terrific site.
Also a thing to mention is that an online business administration program is designed for people to be able to well proceed to bachelors degree programs. The Ninety credit diploma meets the other bachelor education requirements and when you earn your own associate of arts in BA online, you will possess access to the most up-to-date technologies in this field. Some reasons why students have to get their associate degree in business is because they are interested in this area and want to receive the general knowledge necessary ahead of jumping into a bachelor diploma program. Many thanks for the tips you actually provide within your blog.
Aw, this was a very nice post. In thought I want to put in writing like this moreover ? taking time and actual effort to make an excellent article? however what can I say? I procrastinate alot and not at all seem to get one thing done.
Oh my goodness! an amazing article. Great work.
You have some helpful ideas! Maybe I should consider doing this by myself.
I like your style!
Hey very cool site!! Man .. Beautiful .. Amazing .. I will bookmark your website and take the feeds also…I’m happy to find so many useful information here in the post, we need develop more strategies in this regard, thanks for sharing. . . . . .
It?s laborious to search out knowledgeable individuals on this subject, but you sound like you understand what you?re talking about! Thanks
How long does it take you to write an article like this?
Pretty nice post. I just stumbled upon your weblog and wanted to say that I’ve really enjoyed surfing around your blog posts. After all I’ll be subscribing in your feed and I am hoping you write again very soon!
you have an amazing blog right here! would you prefer to make some invite posts on my blog?
I really believe you will do well in the future I appreciate everything you have added to my knowledge base.
Its like you read my mind! You appear to know a lot about this, like you wrote the book in it or something. I think that you can do with some pics to drive the message home a little bit, but instead of that, this is excellent blog. A fantastic read. I’ll certainly be back.
This article actually helped me with a report I was doing.
Its like you read my thoughts! You seem to know a lot about this, such as you wrote the book in it or something. I believe that you simply could do with a few to pressure the message house a little bit, however other than that, that is magnificent blog. A great read. I’ll certainly be back.
cheap viagra
Thanks for your write-up on the vacation industry. I’d personally also like to add that if you are one senior considering traveling, it really is absolutely important to buy travel insurance for golden-agers. When traveling, retirees are at biggest risk of getting a healthcare emergency. Obtaining the right insurance coverage package for the age group can protect your health and give you peace of mind.
Hey there are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and set up my own. Do you need any html coding expertise to make your own blog? Any help would be really appreciated!
Hey! awesome blog! I happen to be a daily visitor to your site (somewhat more like addict 😛 ) of this website. Just wanted to say I appreciate your blogs and am looking forward for more!
you’re really a good webmaster. The web site loading speed is amazing. It seems that you are doing any unique trick. Moreover, The contents are masterpiece. you’ve done a wonderful job on this topic!
WONDERFUL Post.thanks for share..extra wait .. ?
Nice Post. It’s really a very good article. I noticed all your important points. Thanks.
so much superb information on here, : D.
Thank you for another informative site. Where else could I get that type of information written in such an ideal way? I have a project that I’m just now working on, and I’ve been on the look out for such info.
Normally I do not read article on blogs, but I would like to say that this write-up very forced me to try and do so! Your writing style has been amazed me. Thanks, very nice article.
Déterminer quel casino en ligne est réellement adapté pour miser de l’argent véritable s’avère bien plus délicat qu’on pourrait le penser. En 2025, le secteur français des jeux virtuels connaît une croissance rapide, mettant à disposition plus de 200 plateformes certifiées accessibles aux joueurs nationaux. Cette multitude d’options rend la sélection d’un site fiable et agréable encore plus exigeante.C’est pour cette raison que notre classement s’appuie sur une analyse rigoureuse. L’ensemble des casinos en argent réel mentionnés ici ont été examinés en détail avant d’être ajoutés. La sécurité des utilisateurs demeure au cœur de nos priorités, et notre objectif est de vous éviter les dangers associés aux sites non fiables, de plus en plus nombreux sur le web.
Top 9 Meilleur casino en ligne pour les joueurs français: Winamax – idéal pour le poker et les tournois en ligne, Pokerstars – top pour affronter les joueurs de poker du monde entier, Wazamba – parfait pour les tournois réguliers attractifs, PMU – spécialisé sur le turf, mais propose aussi les paris sportifs, Unibet – idéal pour varier les jeux en ligne, FDJ – meilleure option pour les jeux de loterie et les jeux traditionnels français, Betclic – offre une interface intuitive et des paris combinés, Parionsweb – pensé pour les parieurs français traditionnels, Bwin – intéressant pour les paris sportifs et options hybrides. Le marché du casino en ligne en France connaît une croissance exceptionnelle en 2025. Avec plus de 3 millions de joueurs actifs, les casinos en ligne français offrent une expérience de jeu de plus en plus sophistiquée et sécurisée. Ce guide complet vous présente les meilleurs casinos en ligne disponibles en France, leurs bonus exclusifs, et toutes les informations essentielles pour jouer en toute sécurité.
I used to be able to find good information from your blog posts.
hello!,I like your writing very much! share we communicate more about your article on AOL? I need an expert on this area to solve my problem. May be that’s you! Looking forward to see you.
I have realized that online diploma is getting common because getting your degree online has changed into a popular option for many people. A large number of people have not really had an opportunity to attend an established college or university nonetheless seek the improved earning possibilities and a better job that a Bachelor’s Degree offers. Still other folks might have a diploma in one course but would like to pursue something they now have an interest in.
Another issue is that video games are generally serious in nature with the principal focus on mastering rather than entertainment. Although, there’s an entertainment element to keep your kids engaged, every game is usually designed to improve a specific expertise or program, such as mathmatical or scientific discipline. Thanks for your publication.
Good day! I could have sworn I’ve been to this site before but after going through a few of the articles I realized it’s new to me. Regardless, I’m certainly happy I discovered it and I’ll be bookmarking it and checking back regularly.
Hello there, I found your website by the use of Google whilst looking for a comparable matter, your site got here up, it appears to be like good. I have bookmarked it in my google bookmarks.
Can I simply just say what a comfort to discover an individual who really understands what they are talking about on the net. You definitely understand how to bring an issue to light and make it important. More people must check this out and understand this side of the story. I can’t believe you aren’t more popular because you certainly have the gift.
I have noticed that over the course of creating a relationship with real estate entrepreneurs, you’ll be able to get them to understand that, in each and every real estate exchange, a fee is paid. Finally, FSBO sellers never “save” the fee. Rather, they struggle to win the commission through doing the agent’s job. In the process, they shell out their money and time to accomplish, as best they will, the assignments of an real estate agent. Those responsibilities include revealing the home by means of marketing, showing the home to all buyers, making a sense of buyer emergency in order to trigger an offer, preparing home inspections, managing qualification investigations with the loan company, supervising fixes, and assisting the closing of the deal.
Next time I read a blog, I hope that it won’t fail me just as much as this one. After all, Yes, it was my choice to read through, however I actually thought you’d have something helpful to say. All I hear is a bunch of crying about something you can fix if you were not too busy looking for attention.
Sometimes, the sheer magnitude of the information seems overwhelming.
With CatchON TV, you’ll enjoy the ultimate TV experience — access over 24000 live channels and 90000+ movies and series. Stream everything you love, …
A colleague in the field told me to check out your website.
Abnormal this put up is totaly unrelated to what I was searching google for, but it surely used to be listed at the first page. I suppose your doing one thing proper if Google likes you adequate to place you at the first page of a non similar search.
I am glad to be a visitor of this thoroughgoing web blog ! , regards for this rare information! .
These are some of the most important issues we’ll face over the next few decades.
I really like it when folks come together and share views. Great site, keep it up.
This will be helpful for my family.
Can I simply just say what a relief to find somebody that truly knows what they are talking about on the web. You certainly realize how to bring an issue to light and make it important. More people really need to look at this and understand this side of the story. I can’t believe you aren’t more popular because you surely have the gift.
You need to be a part of a contest for one of the best sites on the internet. I will recommend this website!
You really make it seem so easy with your presentation but I find this topic to be really something which I think I would never understand. It seems too complex and extremely broad for me. I am looking forward for your next post, I?ll try to get the hang of it!
Thanks for the helpful content. It is also my belief that mesothelioma cancer has an very long latency period of time, which means that signs of the disease might not exactly emerge right until 30 to 50 years after the first exposure to mesothelioma. Pleural mesothelioma, that is certainly the most common form and has an effect on the area throughout the lungs, might cause shortness of breath, upper body pains, plus a persistent cough, which may result in coughing up bloodstream.
I’ll check back after you publish more articles.
Very good post. I will be going through some of these issues as well..
Your posts provide a clear, concise description of the issues.
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You clearly know what youre talking about, why waste your intelligence on just posting videos to your blog when you could be giving us something informative to read?
Good post. I study something more challenging on different blogs everyday. It’ll at all times be stimulating to learn content material from other writers and practice just a little one thing from their store. I?d want to make use of some with the content material on my weblog whether you don?t mind. Natually I?ll offer you a link on your net blog. Thanks for sharing.
ho88 là nhà cái cá cược trực tuyến hàng đầu, cung cấp cá cược thể thao, casino, bắn cá với tỷ lệ cược cao, bảo mật tối ưu và nhiều ưu đãi hấp dẫn.
You are one talented writer thank you for the post.
Hey, I simply hopped over to your website by way of StumbleUpon. No longer one thing I’d normally learn, but I preferred your thoughts none the less. Thanks for making one thing worth reading.
Your thing regarding creating will be practically nothing in short supply of awesome. This informative article is incredibly useful and contains offered myself a better solution to be able to my own issues. Which can be the specific purpose MY PARTNER AND I has been doing a search online. I am advocating this informative article with a good friend. I know they are going to get the write-up since beneficial as i would. Yet again many thanks.
Thanks a lot for the helpful content. It is also my belief that mesothelioma has an incredibly long latency time period, which means that signs and symptoms of the disease would possibly not emerge right until 30 to 50 years after the 1st exposure to asbestos. Pleural mesothelioma, that is the most common style and impacts the area throughout the lungs, will cause shortness of breath, breasts pains, plus a persistent coughing, which may result in coughing up our blood.
Nice post. I study one thing tougher on totally different blogs everyday. It will at all times be stimulating to learn content material from other writers and practice a little bit one thing from their store. I’d prefer to use some with the content material on my blog whether you don’t mind. Natually I’ll provide you with a hyperlink in your net blog. Thanks for sharing.
I just sent this post to a bunch of my friends as I agree with most of what you’re saying here and the way you’ve presented it is awesome.
hello!,I like your writing very much! share we communicate more about your post on AOL? I require a specialist on this space to unravel my problem. May be that’s you! Looking forward to see you.
I really like what you guys are up too. This type of clever work and exposure! Keep up the great works guys I’ve included you guys to my blogroll.
okmark your weblog and check again here frequently. I’m quite sure I will learn plenty of new stuff right here! Best of luck for the next!
That is a good tip especially to those fresh to the blogosphere. Short but very accurate information… Thank you for sharing this one. A must read article!
Having read this I believed it was extremely informative. I appreciate you taking the time and effort to put this informative article together. I once again find myself personally spending a significant amount of time both reading and posting comments. But so what, it was still worth it!
Next time I read a blog, I hope that it won’t disappoint me as much as this particular one. I mean, Yes, it was my choice to read, but I really thought you would have something helpful to talk about. All I hear is a bunch of crying about something you could possibly fix if you were not too busy seeking attention.
It’s hard to come by knowledgeable people for this topic, however, you sound like you know what you’re talking about! Thanks
I have to disagree with most of the comments here, but maybe I’m just a contrarian.
That is a great tip especially to those new to the blogosphere. Simple but very accurate info… Many thanks for sharing this one. A must read article.
Oh my goodness! Incredible article dude! Thank you so much, However I am going through problems with your RSS. I don’t know the reason why I can’t subscribe to it. Is there anybody having similar RSS problems? Anybody who knows the solution can you kindly respond? Thanks!!
Thanks for your patience and sorry for the inconvenience!
I was reading through some of your content on this internet site and I believe this web site is very informative ! Continue posting .
Very good info. Lucky me I ran across your blog by chance (stumbleupon). I have saved as a favorite for later!
Thanks for any other informative web site. The place else could I am getting that type of information written in such an ideal approach? I’ve a project that I’m just now working on, and I’ve been on the glance out for such information.
Howdy! I simply wish to give you a big thumbs up for the excellent info you have right here on this post. I will be returning to your site for more soon.
I have seen a great deal of useful factors on your web page about desktops. However, I have got the view that notebook computers are still less than powerful more than enough to be a wise decision if you often do tasks that require plenty of power, for example video touch-ups. But for world wide web surfing, word processing, and a lot other common computer functions they are just fine, provided you do not mind the tiny screen size. Thanks for sharing your thinking.
I was pretty pleased to find this great site. I need to to thank you for your time due to this wonderful read!! I definitely liked every part of it and I have you book marked to see new stuff in your web site.
You could definitely see your skills in the work you write. The world hopes for more passionate writers like you who aren’t afraid to say how they believe. Always follow your heart.
you’re in reality a just right webmaster. The web site loading velocity is incredible. It sort of feels that you’re doing any distinctive trick. In addition, The contents are masterpiece. you’ve performed a great process on this topic!
I’m impressed, I have to admit. Seldom do I encounter a blog that’s both equally educative and engaging, and let me tell you, you’ve hit the nail on the head. The issue is something which too few people are speaking intelligently about. I’m very happy that I found this in my search for something regarding this.
Great post. I was checking continuously this weblog and I’m impressed! Extremely useful information specially the remaining part 🙂 I deal with such info a lot. I used to be looking for this certain info for a very long time. Thanks and best of luck.
After I originally left a comment I appear to have clicked on the -Notify me when new comments are added- checkbox and now whenever a comment is added I receive four emails with the exact same comment. There has to be a way you are able to remove me from that service? Cheers.
I think other web site proprietors should take this website as an model, very clean and fantastic user genial style and design, as well as the content. You are an expert in this topic!
This site was… how do you say it? Relevant!! Finally I’ve found something which helped me. Cheers.
Wonderful beat ! I wish to apprentice while you amend your site, how could i subscribe for a blog site? The account helped me a acceptable deal. I had been tiny bit acquainted of this your broadcast offered bright clear idea
I’m blown away by the quality of this content! The author has undoubtedly put a huge amount of effort into investigating and organizing the information. It’s refreshing to come across an article that not only gives valuable information but also keeps the readers engaged from start to finish. Kudos to him for creating such a masterpiece!
Kèo nhà cái bóng đá hôm nay mới nhất. Xem tỷ lệ kèo nhà cái 5 trực tuyến tối đêm nay chuẩn xác. Tỷ lệ kèo cá cược keonhacai 55 trực tiếp nhanh nhất 24h qua.
Everything is very open with a precise description of the challenges. It was definitely informative. Your website is extremely helpful. Many thanks for sharing!
Nice post. I study something more challenging on completely different blogs everyday. It should all the time be stimulating to learn content material from other writers and apply just a little something from their store. I?d desire to use some with the content on my blog whether or not you don?t mind. Natually I?ll offer you a hyperlink on your net blog. Thanks for sharing.
Spot on with this write-up, I truly think this website needs rather more consideration. I?ll in all probability be once more to learn much more, thanks for that info.
This is a good tip especially to those new to the blogosphere. Brief but very precise info… Appreciate your sharing this one. A must read post!
Link tải về toàn file nén có mã độc tống tiền ransomware.
Mẹ kiếp mất tiền đã đằng này còn bị lấy thông tin đi vay nợ hộ.
Nó còn bán dữ liệu người xem cho mấy đường dây ngầm kiếm tiền bẩn.
Click vào quảng cáo của nó là tự động tải về một đống phần mềm gián điệp.
Cái web này là một cái bẫy. Vào là chỉ có mất tiền và thông tin.
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You obviously know what youre talking about, why throw away your intelligence on just posting videos to your blog when you could be giving us something informative to read?
Click vào quảng cáo của nó là tự động tải về một đống phần mềm gián điệp.
Tránh xa cái ổ lừa đảo này ra nếu không muốn tan nhà nát cửa.
Attractive section of content. I just stumbled upon your weblog and in accession capital to assert that I get in fact enjoyed account your blog posts. Any way I?ll be subscribing to your feeds and even I achievement you access consistently quickly.
After exploring a number of the blog posts on your web site, I really appreciate your way of writing a blog. I saved as a favorite it to my bookmark webpage list and will be checking back soon. Please check out my website too and let me know what you think.
Our family had similar issues, thanks.
My coder is trying to convince me to move to .net from PHP. I have always disliked the idea because of the expenses. But he’s tryiong none the less. I’ve been using WordPress on numerous websites for about a year and am nervous about switching to another platform. I have heard great things about blogengine.net. Is there a way I can import all my wordpress posts into it? Any help would be really appreciated!
You need to really control the comments listed here
Good day! I could have sworn I’ve been to this web site before but after browsing through many of the posts I realized it’s new to me. Nonetheless, I’m certainly happy I discovered it and I’ll be bookmarking it and checking back regularly!
Hi, I do think this is an excellent site. I stumbledupon it 😉 I will revisit yet again since i have bookmarked it. Money and freedom is the greatest way to change, may you be rich and continue to help others.
An outstanding share! I’ve just forwarded this onto a co-worker who was conducting a little homework on this. And he actually bought me dinner due to the fact that I stumbled upon it for him… lol. So allow me to reword this…. Thank YOU for the meal!! But yeah, thanx for spending time to discuss this issue here on your website.
Thanks for the suggestions shared in your blog. One more thing I would like to mention is that weight loss is not all about going on a dietary fad and trying to reduce as much weight as you’re able in a couple of days. The most effective way to lose weight naturally is by taking it slowly but surely and obeying some basic tips which can enable you to make the most out of your attempt to slim down. You may be aware and be following some of these tips, nevertheless reinforcing knowledge never hurts.
Good article! We are linking to this particularly great content on our site. Keep up the great writing.
Hello there! I could have sworn I’ve visited this web site before but after looking at many of the articles I realized it’s new to me. Anyways, I’m definitely happy I found it and I’ll be book-marking it and checking back regularly!
Oh my goodness! Impressive article dude! Thanks, However I am having issues with your RSS. I don’t know why I am unable to join it. Is there anybody getting identical RSS issues? Anyone that knows the solution can you kindly respond? Thanks!!
Greetings! Very helpful advice within this post! It is the little changes that produce the most significant changes. Thanks a lot for sharing!
I believe you have remarked on some very interesting points , thankyou for the post.
I was able to find good advice from your blog posts.
After I originally commented I seem to have clicked the -Notify me when new comments are added- checkbox and from now on every time a comment is added I get 4 emails with the exact same comment. There has to be a way you are able to remove me from that service? Thanks.
Its like you read my mind! You seem to know so much about this, like you wrote the book in it or something. I think that you could do with a few pics to drive the message home a little bit, but instead of that, this is great blog. A fantastic read. I’ll definitely be back.
Might we expect to see more of these same problems in the future?
bookmarked!!, I love your site!|
Hello! I could have sworn I’ve been to this website before but after reading through some of the post I realized it’s new to me. Nonetheless, I’m definitely glad I found it and I’ll be book-marking and checking back often!
Some genuinely choice content on this site, bookmarked .
Write more stories, more chapters.
Do you offer workshops?
I think it is a nice point of view. I most often meet people who rather say what they suppose others want to hear. Good and well written! I will come back to your site for sure!
Well done! Keep up this quality!
I cannot thank you more than enough for the blogposts on your website. I know you set a lot of time and energy into these and truly hope you know how deeply I appreciate it. I hope I’ll do a similar thing person sooner or later.
of course like your web-site however you have to check the spelling on several of your posts. Many of them are rife with spelling problems and I find it very troublesome to tell the reality then again I will surely come back again.
I want to to thank you for this fantastic read!! I absolutely loved every bit of it. I have got you book marked to check out new things you post…
Great blog right here! You seem to put a significant amount of material on the site rather quickly.
Nice read, I just passed this onto a colleague who was doing some research on that. And he just bought me lunch as I found it for him smile Therefore let me rephrase that: Thank you for lunch!
bien life
Amazing! This blog looks just like my old one! It’s on a totally different topic but it has pretty much the same page layout and design. Outstanding choice of colors!
I think the admin of this website is actually working hard in favor of his web site, since here every material is quality based data.|
You are not right. I am assured. I can prove it. Write to me in PM, we will talk.
I’ve thought about posting something about this before. Good job! Can I use part of your post in my blog?
nicee content keep writing
Pretty! This has been an incredibly wonderful article. Many thanks for providing these details.
After I initially left a comment I appear to have clicked the -Notify me when new comments are added- checkbox and from now on every time a comment is added I receive four emails with the same comment. Perhaps there is a way you can remove me from that service? Cheers.
Good day! I just would like to give an enormous thumbs up for the nice data you will have here on this post. I shall be coming back to your weblog for extra soon.
Pretty! This has been an extremely wonderful article. Many thanks for supplying this info.
My spouse and I stumbled over here coming from a different page and thought I might as well check things out. I like what I see so now i am following you. Look forward to checking out your web page again.
I blog frequently and I genuinely thank you for your information. This article has truly peaked my interest. I will take a note of your site and keep checking for new information about once a week. I opted in for your RSS feed too.
Substantially, the post is really the best on this laudable topic. I concur with your conclusions and will eagerly watch forward to your future updates.Just saying thanx will not just be enough, for the wonderful lucidity in your writing.
After I initially left a comment I appear to have clicked the -Notify me when new comments are added- checkbox and from now on each time a comment is added I get 4 emails with the exact same comment. There has to be a way you can remove me from that service? Kudos.
Everyone loves it when individuals come together and share thoughts. Great site, stick with it!
bookmarked!!, I love your site!
I was able to find good information from your content.
There is definately a lot to find out about this subject. I really like all of the points you made.
You should be a part of a contest for one of the highest quality websites on the web. I most certainly will recommend this blog!
I am glad to talk with you and you give me great help
Great post! We will be linking to this great content on our website. Keep up the good writing.
It is perfect time to make some plans for the future and it is time to be happy. I’ve read this post and if I could I want to suggest you some interesting things or suggestions. Perhaps you can write next articles referring to this article. I wish to read more things about it!
Good information. Lucky me I recently found your blog by chance (stumbleupon). I’ve bookmarked it for later!
Awesome post. It’s so good to see someone taking the time to share this information
Our local network of agencies has found your research so helpful.
Some truly nice stuff on this website , I like it.
Nhìn chuyên nghiệp vậy thôi chứ bên trong toàn lừa đảo, dụ dỗ đầu tư vớ vẩn.
Dịch vụ khách hàng cái đéo gì, lừa được tiền xong là nó block mình luôn.
Né gấp! Share link tải phần mềm có dính virus, cài vào là mất hết dữ liệu.
I blog often and I genuinely appreciate your content. This great article has truly peaked my interest. I am going to book mark your blog and keep checking for new information about once per week. I opted in for your Feed too.
This site was… how do I say it? Relevant!! Finally I’ve found something that helped me. Many thanks.
Greetings! Very useful advice in this particular article! It is the little changes that produce the biggest changes. Thanks a lot for sharing!
Beneficial Blog! I had been simply just debating that there are plenty of screwy results at this issue you now purely replaced my personal belief. Thank you an excellent write-up.
Το στοίχημα αποτελεί αναπόσπαστο κομμάτι της ψυχαγωγίας για πολλούς Έλληνες παίκτες. Καθώς η εγχώρια αγορά υπόκειται σε αυστηρή ρύθμιση και περιορισμούς, ένα μεγάλο μέρος των παικτών στρέφεται προς ξένες στοιχηματικές εταιρείες που προσφέρουν μεγαλύτερη ευελιξία, καλύτερες αποδόσεις, περισσότερες επιλογές και καινοτόμα χαρακτηριστικά. Ειδικά για όσους αναζητούν μια πιο πλούσια στοιχηματική εμπειρία, οι πλατφόρμες του εξωτερικού προσφέρουν σημαντικά πλεονεκτήματα. Ο όρος ξένες στοιχηματικές εταιρείες που δέχονται Έλληνες δεν αφορά απαραίτητα αδειοδοτημένους παρόχους στην Ελλάδα, αλλά ιστοσελίδες που λειτουργούν νόμιμα υπό διεθνείς άδειες όπως της Κουρασάο, της Ανζουάν ή της Κόστα Ρίκα και επιτρέπουν εγγραφή και στοιχηματισμό από Έλληνες χρήστες. Το ενδιαφέρον για τέτοιου είδους εταιρείες αυξάνεται συνεχώς, καθώς προσφέρουν όχι μόνο καλύτερα μπόνους και προωθητικές ενέργειες, αλλά και υποστήριξη σε κρυπτονομίσματα, μηδενική γραφειοκρατία και πιο φιλικές προς τον χρήστη εμπειρίες.
I’m very happy to uncover this page. I want to to thank you for ones time just for this wonderful read!! I definitely appreciated every part of it and I have you book marked to look at new things in your web site.
of course like your web-site however you have to check the spelling on several of your posts. Many of them are rife with spelling problems and I find it very troublesome to tell the reality then again I will surely come back again.
Hi, I just hopped over to your web-site through StumbleUpon. Not somthing I might typically browse, but I liked your views none the less. Thanks for making something worthy of reading through.
Simply want to say your article is as amazing. The clarity in your post is just great and i can assume you’re an expert on this subject. Fine with your permission allow me to grab your feed to keep up to date with forthcoming post. Thanks a million and please keep up the gratifying work.
whoah this blog is excellent i love reading your posts. Keep up the good work! You know, many people are searching around for this info, you can help them greatly.
Toàn nội dung rác, câu view bẩn thỉu, mục đích là lùa gà.
Địt mẹ nó, tao vừa bị hack mất cái nick Facebook sau khi đăng ký ở đây.
I imagine so. Very good stuff, I agree totally.
Terrific work! This is the type of info that should be shared across the net. Shame on Google for not positioning this put up upper! Come on over and visit my website . Thank you =)
This is the right site for anybody who really wants to find out about this topic. You realize a whole lot its almost tough to argue with you (not that I actually would want to…HaHa). You definitely put a fresh spin on a subject that’s been written about for years. Great stuff, just great.
That is a really good tip especially to those new to the blogosphere. Brief but very precise information… Thank you for sharing this one. A must read post.
This is a topic which is near to my heart… Take care! Exactly where can I find the contact details for questions?
My coder is trying to convince me to move to .net from PHP. I have always disliked the idea because of the expenses. But he’s tryiong none the less. I’ve been using WordPress on numerous websites for about a year and am nervous about switching to another platform. I have heard great things about blogengine.net. Is there a way I can import all my wordpress posts into it? Any help would be really appreciated!
Substantially, the post is really the best on this laudable topic. I concur with your conclusions and will eagerly watch forward to your future updates.Just saying thanx will not just be enough, for the wonderful lucidity in your writing.
Attractive portion of content. I simply stumbled upon your site and in accession capital to assert that I get actually loved account your blog posts. Any way I?ll be subscribing to your feeds or even I achievement you get admission to persistently fast.
I do believe all of the ideas you’ve offered to your post. They are very convincing and will definitely work. Nonetheless, the posts are too short for beginners. May you please prolong them a little from next time? Thanks for the post.
Just want to say what a great blog you got here!I’ve been around for quite a lot of time, but finally decided to show my appreciation of your work!
Way cool! Some extremely valid points! I appreciate you penning this post and the rest of the website is really good.
I am constantly invstigating online for posts that can benefit me. Thank you!
Everyone loves what you guys are up too. This kind of clever work and exposure! Keep up the great works guys I’ve you guys to blogroll.|
Terrific post however , I was wondering if you could write a litte more on this subject? I’d be very thankful if you could elaborate a little bit more. Cheers!
you’ve gotten an ideal weblog right here! would you prefer to make some invite posts on my blog?
Thanks , I’ve recently been searching for info approximately this topic for ages and yours is the greatest I’ve came upon so far. But, what concerning the bottom line? Are you positive in regards to the source?
I?ve learn a few excellent stuff here. Certainly price bookmarking for revisiting. I wonder how so much effort you place to create one of these magnificent informative site.
Hey There. I found your blog using msn. This is a very well written article. I?ll be sure to bookmark it and come back to read more of your useful information. Thanks for the post. I will definitely comeback.
it is a really nice point of view. I usually meet people who rather say what they suppose others want to hear. Good and well written! I will come back to your site for sure!
We can see that we need to develop policies to deal with this trend.
This is my first time i visit here. I found so many helpful stuff in your website especially its discussion. From the tons of responses on your posts, I guess I am not the only one having all the enjoyment here! keep up the excellent work
Our local network of agencies has found your research so helpful.
Right here is the perfect webpage for anyone who hopes to find out about this topic. You know a whole lot its almost hard to argue with you (not that I actually would want to…HaHa). You definitely put a brand new spin on a subject that’s been written about for many years. Great stuff, just excellent.
Thanks for another great post. Where else may anybody get that type of info in such an ideal way of writing? I have a presentation next week, and I’m at the search for such information.
I have to thank you for the efforts you’ve put in writing this blog. I am hoping to view the same high-grade content by you later on as well. In truth, your creative writing abilities has motivated me to get my own blog now 😉
If you desire to grow your know-how just keep visiting this site and be updated with the latest news update posted here.|
It sounds like you’re creating problems yourself by defining this as such a comprehensive, almost unknowable problem. Isn’t that self-defeating?
An impressive share! I have just forwarded this onto a friend who had been conducting a little research on this. And he actually bought me breakfast due to the fact that I discovered it for him… lol. So let me reword this…. Thanks for the meal!! But yeah, thanks for spending time to talk about this subject here on your web site.
This is the right web site for anyone who really wants to understand this topic. You know a whole lot its almost hard to argue with you (not that I personally will need to…HaHa). You definitely put a new spin on a subject which has been discussed for years. Excellent stuff, just great.
I need to to thank you for this excellent read!! I absolutely enjoyed every bit of it. I have you book marked to look at new stuff you post…
I used to be able to find good info from your blog articles.
This article actually helped me with a report I was doing.
I like your style!
My brother suggested I might like this websiteHe was once totally rightThis post truly made my dayYou can not imagine simply how a lot time I had spent for this information! Thanks!
Your thing regarding creating will be practically nothing in short supply of awesome. This informative article is incredibly useful and contains offered myself a better solution to be able to my own issues. Which can be the specific purpose MY PARTNER AND I has been doing a search online. I am advocating this informative article with a good friend. I know they are going to get the write-up since beneficial as i would. Yet again many thanks.
Makes sense to me.
Nevertheless, it’s all carried out with tongues rooted solidly in cheeks, and everybody has got nothing but absolutely love for their friendly neighborhood scapegoat. In reality, he is not merely a pushover. He is simply that extraordinary breed of person solid enough to take all that good natured ribbing for what it really is.
Wow, amazing blog layout! How long have you been blogging for? you made blogging look easy. The overall look of your site is great, as well as the content!
Took me time to read all the comments, but I really enjoyed the article. It proved to be Very helpful to me and I am sure to all the commenters here It’s always nice when you can not only be informed, but also entertained I’m sure you had fun writing this article.
bonjour I love Your Blog can not say I come here often but im liking what i c so far….
Hmm it seems like your blog ate my first comment (it was super long) so I guess I’ll just sum it up what I wrote and say, I’m thoroughly enjoying your blog. I as well am an aspiring blog writer but I’m still new to everything. Do you have any suggestions for rookie blog writers? I’d really appreciate it.
That’s some inspirational stuff. Never knew that opinions might be this varied. Thanks for all the enthusiasm to supply such helpful information here.
You are not right. I am assured. I can prove it. Write to me in PM, we will talk.
I concur with your conclusions and will eagerly look forward to your future updates. The usefulness and significance is overwhelming and has been invaluable to me!
Youre so cool! I dont suppose Ive learn something like this before. So good to search out any individual with some authentic ideas on this subject. realy thanks for beginning this up. this web site is one thing that’s wanted on the net, somebody with somewhat originality. helpful job for bringing one thing new to the internet!
How do I subscribe to your blog? Thanks for your help.
Assume you are doing good linking to position you on the first pages of search engines.
If you don’t mind, where do you host your weblog? I am looking for a very good web host and your webpage seams to be extremely fast and up most the time…
I must thank you for the efforts you have put in penning this site. I am hoping to check out the same high-grade content by you in the future as well. In truth, your creative writing abilities has encouraged me to get my own blog now 😉
Right here is the perfect website for anybody who wants to find out about this topic. You realize so much its almost hard to argue with you (not that I really would want to…HaHa). You definitely put a fresh spin on a topic which has been discussed for a long time. Excellent stuff, just excellent.
If most people wrote about this subject with the eloquence that you just did, I’m sure people would do much more than just read, they act. Great stuff here. Please keep it up.
You have made some good points there. I looked on the web to find out more about the issue and found most people will go along with your views on this website.
This is a topic that’s near to my heart… Best wishes! Exactly where are your contact details though?
I’m impressed, I have to admit. Seldom do I encounter a blog that’s both equally educative and engaging, and without a doubt, you have hit the nail on the head. The issue is something that too few folks are speaking intelligently about. Now i’m very happy that I stumbled across this in my search for something regarding this.
It’s nearly impossible to find well-informed people in this particular topic, but you seem like you know what you’re talking about! Thanks
What’s Happening i am new to this, I stumbled upon this I have found It absolutely helpful and it has helped me out loads. I am hoping to give a contribution & aid different customers like its aided me. Good job.|
I think this is among the so much vital info for me. And i’m happy reading your article. But wanna remark on few common issues, The site style is wonderful, the articles is really excellent : D. Just right job, cheers
Great post. I was checking constantly this blog and I’m impressed! Very useful info specifically the last part 🙂 I care for such info much. I was looking for this certain information for a very long time. Thank you and good luck.
Greetings! This is my first visit to your blog! We are a collection of volunteers and starting a new initiative in a community in the same niche. Your blog provided us beneficial information. You have done a wonderful job!
Magnificent beat ! Can I be your apprentice? Just kidding!
nicee content keep writing
you may have an ideal blog here! would you prefer to make some invite posts on my blog?
Sweet blog! I found it while browsing on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Thank you
One other thing I would like to talk about is that as opposed to trying to accommodate all your online degree training on days of the week that you end work (since most people are fatigued when they come home), try to obtain most of your instructional classes on the saturdays and sundays and only a couple of courses on weekdays, even if it means taking some time away from your weekend. This is fantastic because on the saturdays and sundays, you will be extra rested along with concentrated on school work. Thanks for the different recommendations I have mastered from your blog site.
I know this if off topic but I’m looking into starting my own weblog and was wondering what all is needed to get setup? I’m assuming having a blog like yours would cost a pretty penny? I’m not very web smart so I’m not 100 sure. Any suggestions or advice would be greatly appreciated. Cheers
Its wonderful as your other blog posts : D, regards for putting up.
This is a great tip especially to those new to the blogosphere. Simple but very accurate information… Thank you for sharing this one. A must read article!
I am glad to be a visitor on this website!, regards for this rare information!
The next time I read a blog, Hopefully it won’t fail me just as much as this particular one. I mean, Yes, it was my choice to read through, but I really believed you’d have something helpful to say. All I hear is a bunch of crying about something you could fix if you were not too busy seeking attention.
After I originally commented I seem to have clicked on the -Notify me when new comments are added- checkbox and now every time a comment is added I receive four emails with the exact same comment. Perhaps there is an easy method you are able to remove me from that service? Thanks a lot.
Everything is very open with a really clear clarification of the issues. It was really informative. Your site is extremely helpful. Thank you for sharing!
The post is absolutely fantastic! Lots of great info and inspiration, both of which we all need! Also like to admire the time and effort you put into your website and detailed info you offer! I will bookmark your website!
fantastic post, very informative. I wonder why more of the ther experts in the field do not break it down like this. You should continue your writing. I am confident, you have a great readers’ base already!
I think it is a nice point of view. I most often meet people who rather say what they suppose others want to hear. Good and well written! I will come back to your site for sure!
Hello.This article was extremely interesting, particularly since I was searching for thoughts on this subject last couple of days.
While this issue can vexed most people, my thought is that there has to be a middle or common ground that we all can find. I do value that you’ve added pertinent and sound commentary here though. Thank you!
Greetings! Very useful advice in this particular post! It is the little changes that produce the most important changes. Thanks for sharing!
It’s nearly impossible to find well-informed people for this topic, however, you sound like you know what you’re talking about! Thanks
Hello there! This article couldn’t be written any better! Looking at this post reminds me of my previous roommate! He continually kept preaching about this. I am going to send this post to him. Pretty sure he will have a good read. Thanks for sharing!
Hello to all, it’s really a nice for me to pay a quick visit this site, it consists of valuable Information.|
Hello! I could have sworn I’ve been to your blog before but after looking at some of the posts I realized it’s new to me. Anyhow, I’m certainly delighted I discovered it and I’ll be book-marking it and checking back frequently!
Fantastic piece of writing here1
I blog frequently and I genuinely thank you for your information. This article has truly peaked my interest. I will take a note of your site and keep checking for new details about once a week. I subscribed to your Feed as well.
Hey, I simply hopped over to your website by way of StumbleUpon. No longer one thing I’d normally learn, but I preferred your thoughts none the less. Thanks for making one thing worth reading.
I concur with your conclusions and will eagerly look forward to your future updates. The usefulness and significance is overwhelming and has been invaluable to me!
Nice blog here! Also your web site loads up very fast! What host are you using? Can I get your affiliate link to your host? I wish my web site loaded up as fast as yours lol
I wrote down your blog in my bookmark. I hope that it somehow did not fall and continues to be a great place for reading texts.
I really love your website.. Very nice colors & theme. Did you develop this amazing site yourself? Please reply back as I’m attempting to create my own personal website and would like to learn where you got this from or just what the theme is named. Kudos!
Like the way you’ve outlined things. Easy to follow. Not cluttered.
This site was… how do you say it? Relevant!! Finally I have found something that helped me. Appreciate it.
Good post! We are linking to this great article on our website. Keep up the great writing.
I needed to thank you for this wonderful read!! I definitely enjoyed every bit of it. I have got you book-marked to look at new stuff you post…
I’m impressed, I need to say. Really rarely do I encounter a blog that’s both educational and entertaining, and let me tell you, you have hit the nail on the head.
I love it whenever people come together and share ideas. Great website, continue the good work!|
You have made some really good points there. I looked on the web for more information about the issue and found most people will go along with your views on this site.
Next time I read a blog, Hopefully it does not fail me just as much as this particular one. I mean, Yes, it was my choice to read through, however I actually thought you would probably have something helpful to say. All I hear is a bunch of whining about something that you can fix if you weren’t too busy searching for attention.
Our family had similar issues, thanks.
Hello.This article was extremely interesting, particularly since I was searching for thoughts on this subject last couple of days.
An intriguing discussion is worth comment. There’s no doubt that that you ought to write more about this subject, it may not be a taboo subject but typically people don’t speak about these topics. To the next! Kind regards.
You’re so awesome! I don’t think I have read anything like that before. So great to discover another person with original thoughts on this topic. Really.. thanks for starting this up. This web site is one thing that’s needed on the web, someone with some originality.
Most often since i look for a blog Document realize that the vast majority of blog pages happen to be amateurish. Not so,We can honestly claim for which you writen is definitely great and then your webpage rock solid.
Aw, this was an exceptionally good post. Taking the time and actual effort to generate a superb article… but what can I say… I put things off a whole lot and never manage to get nearly anything done.
Very often I go to see this blog. It very much is pleasant to me. Thanks the author
Enjoyed reading through this, very good material. Thanks!
Spot on with this write-up, I seriously think this site needs far more attention. I’ll probably be returning to read more, thanks for the info!|
Great info! Keep post great articles.
Saw your material, and hope you publish more soon.
What made you first develop an interest in this topic?
Awesome post. It’s so good to see someone taking the time to share this information
Just stumble upon your blog from from time to time. nice article
I’d like to thank you for the efforts you have put in penning this blog. I’m hoping to see the same high-grade content from you in the future as well. In fact, your creative writing abilities has inspired me to get my own, personal site now 😉
Hello there! This article couldn’t be written any better! Looking at this post reminds me of my previous roommate! He constantly kept preaching about this. I most certainly will forward this post to him. Pretty sure he’ll have a very good read. Many thanks for sharing!
It’s difficult to find experienced people in this particular topic, but you sound like you know what you’re talking about! Thanks
I used to be able to find good advice from your blog posts.
Cái web này là một cái bẫy. Vào là chỉ có mất tiền và thông tin.
Đừng có dại mà nhập thông tin thẻ tín dụng vào, nó rút sạch tiền trong tài khoản đấy.
Oh my goodness! Awesome article dude! Thanks, However I am having problems with your RSS. I don’t understand the reason why I can’t join it. Is there anybody having identical RSS issues? Anybody who knows the answer can you kindly respond? Thanx!!
I feel that is among the so much significant info for me. And i am satisfied studying your article. However should commentary on some basic issues, The site style is ideal, the articles is in reality excellent : D. Excellent activity, cheers
I tried a few products from Tillmans Tranquils – indica gummies and really liked the inclusive experience. The gummies contain a cleansed grain, slick texture, and dependable quality. The flavors sensible of consequent, and the portioned servings up it comfortable to choose what works due to the fact that you. Their packaging looks premium and the total feels thoughtfully made. A solid mark with products that are enjoyable and reliable.
I really love your blog.. Very nice colors & theme. Did you develop this site yourself? Please reply back as I’m wanting to create my own personal blog and want to learn where you got this from or exactly what the theme is called. Thanks!
I must thank you for the efforts you’ve put in penning this website. I am hoping to check out the same high-grade blog posts by you in the future as well. In fact, your creative writing abilities has motivated me to get my very own website now 😉
When we look at these issues, we know that they are the key ones for our time.
My brother suggested I might like this websiteHe was once totally rightThis post truly made my dayYou can not imagine simply how a lot time I had spent for this information! Thanks!
Well done! Keep up this quality!
Of course, what a great site and informative posts, I will add backlink – bookmark this site? Regards, Reader
I imagine so. Very good stuff, I agree totally.
I have noticed that credit repair activity really needs to be conducted with techniques. If not, it’s possible you’ll find yourself destroying your rating. In order to reach your goals in fixing your credit history you have to ascertain that from this moment in time you pay your complete monthly dues promptly in advance of their appointed date. It is significant simply because by definitely not accomplishing that, all other methods that you will take to improve your credit rating will not be successful. Thanks for sharing your suggestions.
I think one of your ads triggered my web browser to resize, you might want to put that on your blacklist.
Hello this is kinda of off topic but I was wondering if blogs use WYSIWYG editors or if you have to manually code with HTML. I’m starting a blog soon but have no coding knowledge so I wanted to get advice from someone with experience. Any help would be enormously appreciated!
What a information of un-ambiguity and preserveness of precious familiarity concerning unexpected feelings.|
Hello there, just became aware of your blog through Google, and found that it is truly informative. I am going to watch out for brussels. I will appreciate if you continue this in future. Lots of people will be benefited from your writing. Cheers!
I like meeting utile info, this post has got me even more info!
Spinamba kasyno to dynamiczna przestrzeń rozrywki online, gdzie innowacyjne technologie spotykają się z autentyczną pasją do gier. Nasza platforma łączy w sobie bogatą bibliotekę rozwiązań hazardowych – od ponadczasowych automatów owocowych, przez wyrafinowane gry stołowe, aż po najświeższe wideo sloty sygnowane przez ikony branży: NetEnt, Play’n GO, Pragmatic Play, Microgaming, Evolution Gaming, Betsoft, Booongo, Playson czy Endorphina. Bez względu na to, czy Twoje serce bije dla klasyki z czasów pierwszych kasyn, czy fascynują Cię ultranowoczesne tytuły z mechanizmami Megaways i immeryjną grafiką 3D – u nas odnajdziesz swoją idealną rozgrywkę.
Wybór odpowiedniego kasyna online w Polsce to decyzja, która wymaga uwagi i zrozumienia rynku. W 2026 roku dostępnych jest setki platform hazardowych, ale tylko część z nich oferuje uczciwe warunki, szybkie wypłaty i bezpieczne środowisko gry. Nasz ranking powstał na podstawie szczegółowych testów, analizy licencji, weryfikacji bonusów oraz opinii polskich graczy. Skupiliśmy się na platformach, które akceptują BLIK, oferują polskojęzyczną obsługę i zapewniają transparentne zasady. Każde kasyno w zestawieniu przeszło weryfikację pod kątem bezpieczeństwa, różnorodności gier i jakości obsługi klienta. Dzięki temu możesz podjąć świadomą decyzję i wybrać najlepsze kasyno online, które spełni Twoje oczekiwania.
Nasz portal został stworzony specjalnie dla polskich graczy, którzy szukają rzetelnych informacji o polskie kasyno online. Testujemy i analizujemy polskie kasyna online, które działają na rynku europejskim i akceptują graczy z Polski. Każde polskie kasyno, które opisujemy, przechodzi przez nasz autorski proces weryfikacji – sprawdzamy licencję, ofertę gier, metody płatności, dostępność w języku polskim i zgodność z oczekiwaniami polskich graczy.
Giao diện thì đẹp mà nhân cách thì thối nát, chuyên đi lừa người nhẹ dạ.
Mẹ kiếp, mất tiền thì bực một, bị nó lấy thông tin đi vay app mới cay.
Nội dung toàn xúi bậy, cổ súy cho mấy cái trò phạm pháp.
When I originally left a comment I appear to have clicked on the -Notify me when new comments are added- checkbox and from now on whenever a comment is added I recieve four emails with the same comment. Is there a means you can remove me from that service? Kudos.
Excellent post! We will be linking to this great article on our site. Keep up the great writing.
Excellent blog post. I absolutely appreciate this site. Keep writing!
This is a topic that is close to my heart… Take care! Where can I find the contact details for questions?
You’re so cool! I do not suppose I have read through a single thing like this before. So good to find another person with a few unique thoughts on this topic. Really.. many thanks for starting this up. This site is something that is required on the internet, someone with some originality.
Hi, I do believe this is an excellent website. I stumbledupon it 😉 I will come back once again since i have saved as a favorite it. Money and freedom is the greatest way to change, may you be rich and continue to help others.
With this issue, it’s important to have someone like you with something to say that really matters.
Howdy, I believe your website might be having browser compatibility problems. When I look at your website in Safari, it looks fine however, when opening in Internet Explorer, it’s got some overlapping issues. I just wanted to give you a quick heads up! Besides that, excellent blog!
Hi, I do think this is an excellent web site. I stumbledupon it 😉 I will revisit once again since I bookmarked it. Money and freedom is the greatest way to change, may you be rich and continue to guide others.
Oh my goodness! Impressive article dude! Thank you, However I am having troubles with your RSS. I don’t know the reason why I am unable to join it. Is there anyone else having similar RSS problems? Anyone that knows the solution will you kindly respond? Thanx!
I truly appreciate this post. I have been looking everywhere for this! Thank goodness I found it on Bing. You have made my day! Thx again
After looking at a few of the articles on your site, I really appreciate your technique of blogging. I book-marked it to my bookmark site list and will be checking back soon. Please check out my web site too and tell me how you feel.
Thanks for this wonderful article. Yet another thing to mention is that most digital cameras come equipped with a zoom lens so that more or less of the scene for being included by ‘zooming’ in and out. These types of changes in {focus|focusing|concentration|target|the a**** length usually are reflected inside the viewfinder and on big display screen on the back of your camera.
I have to thank you for the efforts you’ve put in penning this site. I’m hoping to see the same high-grade blog posts by you later on as well. In truth, your creative writing abilities has encouraged me to get my own, personal blog now 😉
Surprisingly good post. I really found your primary webpage and additionally wanted to suggest that have essentially enjoyed searching your website blog posts. Whatever the case I’ll always be subscribing to your entire supply and I hope you jot down ever again soon!
There are some serious financial ramifications here.
Most often since i look for a blog Document realize that the vast majority of blog pages happen to be amateurish. Not so,We can honestly claim for which you writen is definitely great and then your webpage rock solid.
Howdy! I simply wish to give a huge thumbs up for the great information you have here on this post. I will be coming again to your weblog for extra soon.
Spot on with this write-up, I actually assume this website needs far more consideration. I will in all probability be once more to learn rather more, thanks for that info.
This information is critically needed, thanks.
WoW decent article. Can I hire you to guest write for my blog? If so send me an email!
Only a smiling visitor here to share the love (:, btw outstanding style and design .
Oh my goodness! Impressive article dude! Thank you, However I am going through difficulties with your RSS. I don’t know the reason why I can’t join it. Is there anybody getting similar RSS issues? Anyone that knows the solution will you kindly respond? Thanx!!
I’m amazed, I must say. Seldom do I come across a blog that’s both equally educative and amusing, and without a doubt, you’ve hit the nail on the head. The problem is something not enough folks are speaking intelligently about. I am very happy I came across this in my hunt for something concerning this.
An impressive share! I have just forwarded this onto a co-worker who has been conducting a little homework on this. And he in fact bought me breakfast simply because I found it for him… lol. So allow me to reword this…. Thanks for the meal!! But yeah, thanks for spending time to discuss this issue here on your web page.
After exploring a number of the blog posts on your blog, I honestly appreciate your technique of blogging. I book-marked it to my bookmark website list and will be checking back in the near future. Please check out my website too and tell me what you think.
Europe Readr ist mehr als nur eine digitale Bibliothek – es ist ein kulturelles und demokratisches Projekt, das Literatur, Bildung und gesellschaftliches Engagement miteinander verbindet. Initiiert im Rahmen der slowenischen EU-Ratspräsidentschaft im Jahr 2021, bietet Europe Readr eine kostenlose, mehrsprachige Sammlung literarischer Werke aus ganz Europa, die aktuelle gesellschaftliche Themen aufgreifen und zur aktiven Auseinandersetzung anregen.
Keep it up!. I usually don’t post in Blogs but your blog forced me to, amazing work.. beautiful A rise in An increase in An increase in.
I was very pleased to discover this page. I wanted to thank you for ones time for this particularly wonderful read!! I definitely appreciated every bit of it and i also have you saved to fav to look at new information on your website.
Do you offer workshops?
I feel that is among the so much significant info for me. And i am satisfied studying your article. However should commentary on some basic issues, The site style is ideal, the articles is in reality excellent : D. Excellent activity, cheers
Nice post.Very useful info specifically the last part 🙂 Thank you and good luck.
Woah this is just an insane amount of information, must of taken ages to compile so thanx so much for just sharing it with all of us. If your ever in any need of related information, just check out my own site!
Just want to say what a great blog you got here!I’ve been around for quite a lot of time, but finally decided to show my appreciation of your work!
Thanks for the helpful post. It is also my opinion that mesothelioma cancer has an extremely long latency time period, which means that indication of the disease might not emerge until eventually 30 to 50 years after the first exposure to asbestos fiber. Pleural mesothelioma, and that is the most common variety and is affecting the area around the lungs, might cause shortness of breath, upper body pains, including a persistent coughing, which may bring on coughing up body.
We need to build frameworks and funding mechanisms.
Some truly interesting info , well written and broadly user genial .
Hiya, I am really glad I have found this information. Nowadays bloggers publish only about gossip and net stuff and this is actually frustrating.
Very nice article. I definitely appreciate this website. Stick with it!
I’ve thought about posting something about this before. Good job! Can I use part of your post in my blog?
Hi, I do think this is an excellent web site. I stumbledupon it 😉 I may revisit yet again since I saved as a favorite it. Money and freedom is the best way to change, may you be rich and continue to guide others.
Hi, I believe your site might be having web browser compatibility problems. Whenever I take a look at your web site in Safari, it looks fine but when opening in Internet Explorer, it’s got some overlapping issues. I merely wanted to provide you with a quick heads up! Other than that, great website.
Your idea is outstanding; the issue is something that not enough persons are speaking intelligently about. I’m very happy that I stumbled throughout this in my seek for one thing regarding this.
You are one talented writer thank you for the post.
Everyone loves it when people get together and share thoughts. Great site, keep it up!
I was more than happy to find this site. I want to to thank you for ones time due to this fantastic read!! I definitely loved every part of it and I have you bookmarked to see new information on your blog.
A motivating discussion is definitely worth comment. I do believe that you should write more on this topic, it might not be a taboo matter but generally folks don’t speak about such topics. To the next! All the best!
You’re so awesome! I do not think I’ve read through something like this before. So wonderful to discover somebody with some unique thoughts on this issue. Seriously.. many thanks for starting this up. This site is one thing that is needed on the web, someone with a little originality.
It’s hard to come by well-informed people in this particular topic, however, you sound like you know what you’re talking about! Thanks
You need to be a part of a contest for one of the highest quality sites on the net. I most certainly will highly recommend this site!
Hello this is a wonderful write-up. I’m going to e mail this to my friends. I came on this while searching on yahoo I’ll be sure to come back. thanks for sharing.
This has to be one of my favorite posts! And on top of thats its also very helpful topic for newbies. thank a lot for the information!
Bọn này chuyên thu thập thông tin cá nhân rồi đem bán cho tụi đa cấp.
Cái web này là một cái bẫy. Vào là chỉ có mất tiền và thông tin.
Mẹ kiếp, mất tiền thì bực một, bị nó lấy thông tin đi vay app mới cay.
Having read this I believed it was really enlightening. I appreciate you spending some time and energy to put this information together. I once again find myself personally spending a significant amount of time both reading and posting comments. But so what, it was still worthwhile!
Did you remember your main part is wired to respond to CBD? Every magnanimous has an endocannabinoid system (ECS) that influences frame of mind, slumber, and immune response. CBD is unique because it doesn’t swathe immediately to receptors like THC does; in lieu of, it encourages the centre to capitalize on its own endocannabinoids more effectively. It’s like a tune-up in return your biological signaling, promoting efficiency and peace
May I simply just say what a comfort to discover someone who actually knows what they’re talking about on the web. You definitely realize how to bring an issue to light and make it important. More people need to look at this and understand this side of the story. I can’t believe you are not more popular because you definitely possess the gift.
Abnormal this put up is totaly unrelated to what I was searching google for, but it surely used to be listed at the first page. I suppose your doing one thing proper if Google likes you adequate to place you at the first page of a non similar search.
Is it okay to put a portion of this on my weblog if perhaps I post a reference point to this web page?
I have been curious about these trends, and you have really helped me. I have just told a few of my friends about this on FaceBook and they love your content just as much as I do.
Oh my goodness! Impressive article dude! Thank you so much, However I am going through issues with your RSS. I don’t know the reason why I can’t join it. Is there anyone else having similar RSS problems? Anybody who knows the answer can you kindly respond? Thanks.
Does it look like we’re in for a big ride here?
These kind of posts are always inspiring and I prefer to read quality content so I happy to find many good point here in the post. writing is simply wonderful! thank you for the post
I’m so happy to read this. This is the type of manual that needs to be given and not the random misinformation that’s at the other blogs. Appreciate your sharing this best doc.
Good write-up. I certainly love this website. Stick with it!
Very good info. Lucky me I found your site by accident (stumbleupon). I’ve saved it for later!
Way cool! Some very valid points! I appreciate you writing this write-up and also the rest of the website is very good.
You appear to know so much about this, and I see you’re a published author. Thanks
Hey, I simply hopped over to your website by way of StumbleUpon. No longer one thing I’d normally learn, but I preferred your thoughts none the less. Thanks for making one thing worth reading.
I appreciate your work, thanks for all the great blog posts.
whoah this weblog is great i really like studying your articles. Stay up the great work! You already know, lots of persons are looking round for this information, you can aid them greatly.
Admiring the time and effort you put into your site and detailed info you offer!
Thank you, I have just been searching for information about this topic for ages and yours is the greatest I’ve discovered till now. But, what about the conclusion? Are you sure about the source?
A friend of mine advised me to review this site. And yes. it has some useful pieces of info and I enjoyed reading it.
I like this weblog very much so much great info .
An interesting discussion is worth comment. I think that you ought to publish more about this topic, it may not be a taboo matter but usually folks don’t discuss these subjects. To the next! Kind regards.
This is my first time i visit here. I found so many helpful stuff in your website especially its discussion. From the tons of responses on your posts, I guess I am not the only one having all the enjoyment here! keep up the excellent work
I really love this article.
Nice blog. Could someone with little experience do it, and add updates without messing it up? Good information on here, very informative.
Howdy! I could have sworn I’ve been to this website before but after going through some of the posts I realized it’s new to me. Nonetheless, I’m certainly happy I came across it and I’ll be bookmarking it and checking back frequently!
I would really like to appreciate the endeavors you cash in on written this article. I’m going for the similar best product from you finding out in the foreseeable future as well. Actually your creative writing abilities has urged me to begin my very own blog now. Genuinely the blogging is distributing its wings rapidly. Your write down is often a fine illustration showing it.
Good post. I study something more difficult on different blogs everyday. It’s going to always be stimulating to learn content material from other writers and observe a little bit one thing from their store. I’d prefer to use some with the content material on my blog whether you don’t mind. Natually I’ll give you a link in your web blog. Thanks for sharing.
Makes sense to me.
I loved your idea there, I tell you blogs are so exciting sometimes like looking into people’s private life’s and work. Every new remark wonderful in its own right.
Toàn nội dung rác, câu view bẩn thỉu, mục đích là lùa gà.
Usually I don’t learn article on blogs, but I wish to say that this write-up very pressured me to take a look at and do it! Your writing taste has been amazed me. Thanks, quite great post.
Hello. Great job. I did not expect this on a Wednesday. This is a great story. Thanks!
I will share you blog with my sis.
The sketch is tasteful, your authored material stylish.
Nice post.Very useful info specifically the last part 🙂 Thank you and good luck.
I have to say this post was certainly informative and contains useful content for enthusiastic visitors. I will definitely bookmark this website for future reference and further viewing. cheers a bunch for sharing this with us!
Nice blog. Could someone with little experience do it, and add updates without messing it up? Good information on here, very informative.
This blog post is excellent, probably because of how well the subject was developed. I like some of the comments too.
Some truly nice stuff on this website , I like it.
We need to build frameworks and funding mechanisms.
This blog post is excellent, probably because of how well the subject was developed. I like some of the comments too though I could prefer we all stay on the subject in order add value to the subject!
I concur with your conclusions and will eagerly look forward to your future updates. The usefulness and significance is overwhelming and has been invaluable to me!
Hello, i think that i saw you visited my web site so i got here to go back the desire?.I’m trying to find things to enhance my website!I guess its adequate to use some of your concepts!!|
nicee content keep writing
I think other website proprietors should take this web site as an model, very clean and great user pleasant style and design .
If most people wrote about this subject with the eloquence that you just did, I’m sure people would do much more than just read, they act. Great stuff here. Please keep it up.
Hi there! I could have sworn I’ve visited your blog before but after going through many of the posts I realized it’s new to me. Anyhow, I’m definitely pleased I found it and I’ll be bookmarking it and checking back frequently!
nicee content keep writing
Great article. It is rather unfortunate that over the last decade, the travel industry has had to tackle terrorism, SARS, tsunamis, bird flu, swine flu, as well as first ever true global recession. Through all this the industry has proven to be effective, resilient in addition to dynamic, locating new ways to deal with misfortune. There are constantly fresh challenges and opportunity to which the marketplace must once again adapt and respond.
These days of austerity and also relative anxiety about incurring debt, most people balk against the idea of having a credit card to make purchase of merchandise or perhaps pay for any occasion, preferring, instead only to rely on the actual tried along with trusted way of making settlement – cash. However, if you possess the cash on hand to make the purchase entirely, then, paradoxically, that’s the best time to be able to use the cards for several causes.
Most often since i look for a blog Document realize that the vast majority of blog pages happen to be amateurish. Not so,We can honestly claim for which you writen is definitely great and then your webpage rock solid.
very good post, i certainly love this web site, keep on it
That is really fascinating, You’re an excessively skilled blogger. I’ve joined your rss feed and look forward to in the hunt for extra of your magnificent post. Additionally, I’ve shared your website in my social networks!
I don’t know if it’s just me or if everybody else experiencing issues with your site. It appears as though some of the written text on your content are running off the screen. Can someone else please provide feedback and let me know if this is happening to them as well? This could be a problem with my web browser because I’ve had this happen before. Appreciate it
Thank you for all the information was very accurate, just wondering if all this is possible.~
This is a topic that is close to my heart… Cheers! Exactly where can I find the contact details for questions?
Everyone loves it whenever people come together and share views. Great blog, continue the good work!
Way cool! Some extremely valid points! I appreciate you penning this article and the rest of the website is really good.
May I just say what a comfort to discover somebody that actually understands what they are talking about on the web. You definitely understand how to bring an issue to light and make it important. More people should check this out and understand this side of your story. I was surprised that you’re not more popular since you most certainly possess the gift.
I will share you blog with my sis.
I had fun reading this post. I want to see more on this subject.. Gives Thanks for writing this nice article.. Anyway, I’m going to subscribe to your rss and I wish you write great articles again soon.
I will share you blog with my sis.
I saw a similar post on another website but the points were not as well articulated.
Excellent site you have got here.. It’s hard to find quality writing like yours these days. I seriously appreciate people like you! Take care!!
Just wanna admit that this is extremely helpful, Thanks for taking your time to write this.
Glad to be one of many visitants on this amazing site : D.
These are some of the most important issues we’ll face over the next few decades.
I used to be able to find good info from your blog posts.
Sweet blog! I found it while browsing on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Thank you
Excellent site you’ve got here.. It’s difficult to find good quality writing like yours nowadays. I seriously appreciate people like you! Take care!!
Hi, I do believe this is a great blog. I stumbledupon it 😉 I will return once again since i have book-marked it. Money and freedom is the greatest way to change, may you be rich and continue to guide other people.
Way cool! Some extremely valid points! I appreciate you writing this post plus the rest of the website is really good.
Web này có đường dây ngầm, toàn nội dung phi pháp, chính quyền sắp sờ gáy rồi.
I’m impressed, I have to admit. Rarely do I come across a blog that’s both educative and engaging, and without a doubt, you’ve hit the nail on the head. The issue is something not enough people are speaking intelligently about. Now i’m very happy I found this in my search for something regarding this.
When are you going to post again? You really entertain me!
I delight in, cause I discovered exactly what I used to be taking a look for. You’ve ended my four day long hunt! God Bless you man. Have a nice day. Bye
Thanks for your article. I also think that laptop computers have gotten more and more popular today, and now are often the only type of computer used in a household. This is due to the fact that at the same time that they’re becoming more and more cost-effective, their working power is growing to the point where there’re as highly effective as desktop computers from just a few years ago.
Sweet blog! I found it while browsing on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Thank you
Surprisingly good post. I really found your primary webpage and additionally wanted to suggest that have essentially enjoyed searching your website blog posts. Whatever the case I’ll always be subscribing to your entire supply and I hope you jot down ever again soon!
Some genuinely choice content on this site, bookmarked .
Can I just say what a relief to seek out someone who actually knows what theyre speaking about on the internet. You positively know find out how to bring a problem to mild and make it important. Extra individuals have to read this and perceive this side of the story. I cant believe youre not more in style because you positively have the gift.
I would share your post with my sis.
Oh my goodness! an amazing article. Great work.
I would share your post with my sis.
This is one very informative blog. I like the way you write and I will bookmark your blog to my favorites.
Nice Post. It’s really a very good article. I noticed all your important points. Thanks.
Đừng có dại mà nhập thông tin thẻ tín dụng vào, nó rút sạch tiền trong tài khoản đấy.
Dude.. I am not much into reading, but somehow I got to read lots of articles on your blog. Its amazing how interesting it is for me to visit you very often. –
I would share your post with my sis.
Hi there! This post could not be written any better! Looking through this article reminds me of my previous roommate! He constantly kept preaching about this. I am going to forward this article to him. Fairly certain he’ll have a great read. Thank you for sharing!
There is so much to try to understand
Your posts provide a clear, concise description of the issues.
I was very happy to find this website. I want to to thank you for ones time for this fantastic read!! I definitely loved every bit of it and i also have you book-marked to look at new stuff on your site.
I blog often and I seriously thank you for your content. The article has truly peaked my interest. I will take a note of your site and keep checking for new information about once a week. I subscribed to your Feed too.
Yet another thing is that when looking for a good online electronics retail outlet, look for online shops that are regularly updated, keeping up-to-date with the most recent products, the perfect deals, in addition to helpful information on products. This will ensure you are getting through a shop that stays ahead of the competition and provides you things to make knowledgeable, well-informed electronics buys. Thanks for the important tips I’ve learned from your blog.
Can I just say what a relief to discover an individual who actually understands what they’re talking about over the internet. You definitely understand how to bring a problem to light and make it important. More and more people need to read this and understand this side of the story. I was surprised that you are not more popular since you most certainly possess the gift.
How come you do not have your website viewable in mobile format? cant see anything in my Droid.
You are one talented writer thank you for the post.
After checking out a handful of the blog posts on your web site, I honestly appreciate your way of blogging. I book-marked it to my bookmark site list and will be checking back soon. Please visit my web site too and let me know how you feel.
Hi there! I just would like to offer you a big thumbs up for your great info you’ve got right here on this post. I am returning to your site for more soon.
After study a number of of the weblog posts in your web site now, and I actually like your approach of blogging. I bookmarked it to my bookmark web site record and will likely be checking again soon. Pls check out my web site as effectively and let me know what you think.
hello there and thank you for your info ? I?ve certainly picked up anything new from right here. I did however expertise some technical points using this web site, as I experienced to reload the web site a lot of times previous to I could get it to load correctly. I had been wondering if your hosting is OK? Not that I’m complaining, but slow loading instances times will often affect your placement in google and can damage your quality score if advertising and marketing with Adwords. Anyway I?m adding this RSS to my email and could look out for a lot more of your respective intriguing content. Make sure you update this again soon..
Beneficial Blog! I had been simply just debating that there are plenty of screwy results at this issue you now purely replaced my personal belief. Thank you an excellent write-up.
Thanks for your patience and sorry for the inconvenience!
I like your quality that you put into your writing . Please do continue with more like this.
A cool post there mate ! Thank you for posting.
Hi just wanted to give you a brief heads up and let you know a few of the pictures aren’t loading properly. I’m not sure why but I think its a linking issue. I’ve tried it in two different internet browsers and both show the same outcome.
Great post, keep up the good work, I hope you don’t mind but I’ve added on my blog roll.
Somebody necessarily lend a hand to make severely articles I might state. That is the first time I frequented your web page and to this point? I surprised with the analysis you made to create this particular submit amazing. Excellent activity!
I am glad to be a visitor of this perfect blog !, appreciate it for this rare info!
Good job for bringing something important to the internet!
Can I just say what a relief to discover a person that truly knows what they’re talking about online. You definitely realize how to bring a problem to light and make it important. More people really need to look at this and understand this side of your story. It’s surprising you aren’t more popular because you surely have the gift.
I blog often and I seriously thank you for your information. This article has truly peaked my interest. I am going to bookmark your website and keep checking for new information about once per week. I opted in for your Feed as well.
I am glad to be a visitor of this perfect blog !, appreciate it for this rare info!
This information is very important and you’ll need to know this when you constructor your own photo voltaic panel.
Spot on with this write-up, I absolutely think this site needs a great deal more attention. I’ll probably be back again to read through more, thanks for the information.
I really love this article.
Good article. I’m experiencing some of these issues as well..
There is perceptibly a lot to identify about this. I consider you made some good points in features also.
Hello. Great job. I did not expect this on a Wednesday. This is a great story. Thanks!
Thanks for the a new challenge you have unveiled in your text. One thing I would like to reply to is that FSBO associations are built over time. By launching yourself to owners the first saturday their FSBO is usually announced, before the masses get started calling on Monday, you generate a good relationship. By mailing them resources, educational materials, free reports, and forms, you become a great ally. By subtracting a personal affinity for them and their circumstances, you generate a solid network that, many times, pays off if the owners decide to go with a realtor they know along with trust — preferably you.
Tips and tools you offer are so helpful to agencies in our community.
Greetings! This is my first visit to your blog! We are a collection of volunteers and starting a new initiative in a community in the same niche. Your blog provided us beneficial information. You have done a wonderful job!
I’m amazed, I have to admit. Seldom do I come across a blog that’s both educative and entertaining, and without a doubt, you’ve hit the nail on the head. The problem is an issue that too few people are speaking intelligently about. Now i’m very happy I stumbled across this in my search for something relating to this.
There is so much to try to understand
There are some attention-grabbing closing dates on this article however I don?t know if I see all of them middle to heart. There may be some validity but I will take hold opinion until I look into it further. Good article , thanks and we would like more! Added to FeedBurner as well
My developer is trying to persuade me to move to .net from PHP. I have always disliked the idea because of the expenses. But he’s tryiong none the less. I’ve been using WordPress on a variety of websites for about a year and am nervous about switching to another platform. I have heard fantastic things about blogengine.net. Is there a way I can import all my wordpress content into it? Any kind of help would be greatly appreciated!|
It’s the best time to make some plans for the future and it is time to be happy. I’ve read this post and if I could I want to suggest you some interesting issues or suggestions. Maybe you could write next articles relating to this article. I wish to read even more things approximately it!
You can certainly see your expertise in the work you write. The world hopes for even more passionate writers like you who aren’t afraid to say how they believe. Always go after your heart.
I love your blog.. very nice colors & theme. Did you create this website yourself or did you hire someone to do it for you? Plz respond as I’m looking to construct my own blog and would like to find out where u got this from. kudos
This blog was… how do I say it? Relevant!! Finally I’ve found something that helped me. Appreciate it.
Thanks for another great post. Where else could anybody get that kind of info in such a perfect way of writing? I’ve a presentation next week, and I’m on the look for such info.
I’ve been surfing online more than three hours today, yet I never found any interesting article like yours. It is pretty worth enough for me. In my opinion, if all site owners and bloggers made good content as you did, the internet will be a lot more useful than ever before.
There is so much to try to understand
Hello there! I really enjoy reading your blog! If you keep making amazing posts like this I will come back every day to keep reading.
If wings are your thing, Tinker Bell’s sexy Halloween costume design is all grown up.
Assume you are doing good linking to position you on the first pages of search engines.
This is my first time i visit here. I found so many helpful stuff in your website especially its discussion. From the tons of responses on your posts, I guess I am not the only one having all the enjoyment here! keep up the excellent work
Hi there it’s me, I am also visiting this site on a regular basis, this site is really nice and the visitors are in fact sharing good thoughts.|
That’s some inspirational stuff. Never knew that opinions might be this varied. Thanks for all the enthusiasm to supply such helpful information here.
Great post. I was checking constantly this blog and I’m impressed! Very useful info specifically the last part 🙂 I care for such info much. I was looking for this certain information for a very long time. Thank you and good luck.
Thank you pertaining to sharing the following great subject matter on your website. I ran into it on google. I am going to check to come back after you publish additional aricles.
I have been surfing online more than three hours today, yet I never found anything that grabbed my interest as much as this piece.
Woah this is just an insane amount of information, must of taken ages to compile so thanx so much for just sharing it with all of us. If your ever in any need of related information, just check out my own site!
Excellent read, I just passed this onto a colleague who was doing a little research on that. And he actually bought me lunch because I found it for him smile So let me rephrase that.|
Hey very cool site!! Man .. Beautiful .. Amazing .. I will bookmark your website and take the feeds also…I’m happy to find so many useful information here in the post, we need develop more strategies in this regard, thanks for sharing. . . . . .
I like meeting utile info, this post has got me even more info!
I am glad to be one of the visitors on this great site (:, appreciate it for putting up.
Thanks For This Blog, was added to my bookmarks.
What a great article.. i subscribed btw!
Dude.. I am not much into reading, but somehow I got to read lots of articles on your blog. Its amazing how interesting it is for me to visit you very often. –
Hello, I think your blog might be having browser compatibility issues. When I look at your website in Chrome, it looks fine but when opening in Internet Explorer, it has some overlapping. I just wanted to give you a quick heads up! Other than that, awesome blog!
I like what you have to offer. Keep up the good work!
Took me time to read all the comments, but I really enjoyed the article. It proved to be Very helpful to me and I am sure to all the commenters here It’s always nice when you can not only be informed, but also entertained I’m sure you had fun writing this article.
Mình cảm thấy an tâm khi sử dụng website.
Web mang đến trải nghiệm trực quan và dễ hiểu.
Các chức năng trên web vận hành mượt mà, ổn định.
Hello, i think that i saw you visited my web site thus i came to ?return the favor?.I’m attempting to find things to enhance my website!I suppose its ok to use some of your ideas!!
Valuable info. Lucky me I found your website by accident, and I’m shocked why this accident didn’t happened earlier! I bookmarked it.
Web mang lại cảm giác thoải mái khi chơi game.
Nice post.Very useful info specifically the last part 🙂 Thank you and good luck.
Mình cảm thấy thoải mái khi truy cập web.
It’s difficult to find educated people in this particular subject, but you sound like you know what you’re talking about! Thanks
Hi there, just became aware of your blog through Google, and found that it’s truly informative. It’s important to cover these trends.
Great post! We will be linking to this particularly great content on our site. Keep up the good writing.
Web mang lại trải nghiệm chơi game tiện lợi và thú vị.
Mẹ kiếp, mất tiền thì bực một, bị nó lấy thông tin đi vay app mới cay.
Giao diện web hiện đại, bắt mắt và trực quan.
Các danh mục trên web được bố trí hợp lý.
Trải nghiệm chơi game trên web rất mượt mà.
Thông tin trên web rõ ràng, không rối mắt.
Wow, fantastic blog structure! How long have you ever been running a blog for? you make blogging glance easy. The full glance of your site is wonderful, as neatly as the content material!
I’m extremely pleased to discover this website. I wanted to thank you for your time just for this wonderful read!! I definitely liked every part of it and I have you book marked to look at new stuff on your website.
Trò chơi trên web được thiết kế sinh động, bắt mắt.
Mình đánh giá cao các tính năng tiện lợi trên web.
Web giúp thao tác chơi game nhanh chóng và tiện lợi.
magnificent submit, very informative. I ponder why the other experts of this sector do not understand this. You must continue your writing. I’m sure, you have a huge readers’ base already!
Yet another thing is that when looking for a good on the net electronics retail outlet, look for online shops that are constantly updated, preserving up-to-date with the newest products, the most effective deals, as well as helpful information on goods and services. This will ensure that you are doing business with a shop that really stays atop the competition and give you things to make knowledgeable, well-informed electronics buying. Thanks for the critical tips I have really learned through the blog.
Dịch vụ khách hàng cái đéo gì, lừa được tiền xong là nó block mình luôn.
Nội dung toàn xúi bậy, cổ súy cho mấy cái trò phạm pháp.
Pretty! This has been an incredibly wonderful post. Thank you for supplying these details.
It is my belief that mesothelioma is definitely the most lethal cancer. It contains unusual attributes. The more I really look at it the more I am confident it does not respond like a true solid human cancer. If perhaps mesothelioma is really a rogue virus-like infection, hence there is the potential for developing a vaccine plus offering vaccination for asbestos open people who are vulnerable to high risk with developing future asbestos associated malignancies. Thanks for sharing your ideas for this important health issue.
Hi there! This is my first visit to your blog! We are a group of volunteers and starting a new initiative in a community in the same niche. Your blog provided us valuable information to work on. You have done a wonderful job!
The other day, while I was at work, my sister stole my iPad and tested to see if it can survive a 30 foot drop, just so she can be a youtube sensation. My iPad is now broken and she has 83 views. I know this is completely off topic but I had to share it with someone!
At this time it appears like Drupal is the preferred blogging platform out there right now. (from what I’ve read) Is that what you’re using on your blog?
Hello there! This post could not be written any better! Reading this post reminds me of my old room mate! He always kept chatting about this. I will forward this article to him. Fairly certain he will have a good read. Thanks for sharing!
May I just say what a relief to uncover a person that truly understands what they are talking about on the web. You definitely know how to bring an issue to light and make it important. More people need to look at this and understand this side of the story. I was surprised you aren’t more popular because you surely have the gift.
very good publish, i actually love this web site, keep on it
It’s my opinion that a property foreclosure can have a significant effect on the applicant’s life. Property foreclosures can have a Seven to few years negative affect on a borrower’s credit report. A borrower who have applied for a mortgage or almost any loans even, knows that your worse credit rating is, the more challenging it is to get a decent bank loan. In addition, it might affect any borrower’s power to find a decent place to let or hire, if that turns into the alternative housing solution. Thanks for your blog post.
This blog was… how do you say it? Relevant!! Finally I’ve found something that helped me. Kudos!
Hi there! Someone in my Facebook group shared this website with us so I came to give it a look. I’m definitely enjoying the information. I’m book-marking and will be tweeting this to my followers! Outstanding blog and terrific style and design.
I was just seeking this information for some time. After six hours of continuous Googleing, at last I got it in your site. I wonder what’s the lack of Google strategy that don’t rank this type of informative sites in top of the list. Generally the top web sites are full of garbage.
I’d like to thank you for the efforts you have put in penning this blog. I am hoping to view the same high-grade blog posts from you in the future as well. In truth, your creative writing abilities has encouraged me to get my very own site now 😉
I’ve learned several important things through your post. I would also like to express that there may be situation in which you will obtain a loan and do not need a co-signer such as a Government Student Aid Loan. But when you are getting a borrowing arrangement through a standard lender then you need to be ready to have a co-signer ready to allow you to. The lenders may base any decision on the few aspects but the biggest will be your credit worthiness. There are some loan merchants that will as well look at your work history and decide based on that but in many cases it will hinge on your scores.
Superb blog! Do you have any tips for aspiring writers? I’m planning to start my own website soon but I’m a little lost on everything. Would you recommend starting with a free platform like WordPress or go for a paid option? There are so many choices out there that I’m totally confused .. Any tips? Thank you!
I have learned a few important things by means of your post. I will also like to convey that there might be situation where you will get a loan and do not need a cosigner such as a Federal Student Support Loan. But if you are getting a loan through a standard finance company then you need to be able to have a co-signer ready to help you. The lenders will certainly base any decision on the few factors but the biggest will be your credit history. There are some loan companies that will additionally look at your job history and determine based on that but in almost all cases it will be based on on your score.
Thanks for your useful post. Through the years, I have come to understand that the symptoms of mesothelioma are caused by the actual build up of fluid between your lining in the lung and the upper body cavity. The ailment may start within the chest spot and spread to other limbs. Other symptoms of pleural mesothelioma cancer include fat loss, severe deep breathing trouble, fever, difficulty taking in food, and irritation of the neck and face areas. It ought to be noted that some people existing with the disease usually do not experience any serious symptoms at all.
Đừng có dại mà nhập thông tin thẻ tín dụng vào, nó rút sạch tiền trong tài khoản đấy.
Mẹ kiếp, mất tiền thì bực một, bị nó lấy thông tin đi vay app mới cay.
I’m very pleased to uncover this great site. I need to to thank you for your time just for this fantastic read!! I definitely enjoyed every part of it and I have you saved to fav to check out new things in your website.
I loved as much as you’ll receive carried out right here. The sketch is attractive, your authored material stylish. nonetheless, you command get bought an impatience over that you wish be delivering the following. unwell unquestionably come further formerly again as exactly the same nearly very often inside case you shield this increase.
An impressive share! I’ve just forwarded this onto a friend who was conducting a little research on this. And he in fact bought me dinner because I discovered it for him… lol. So allow me to reword this…. Thanks for the meal!! But yeah, thanks for spending some time to talk about this topic here on your web site.
Web cung cấp nhiều lựa chọn trò chơi phù hợp với mọi sở thích.
Hi, Neat post. There’s a problem with your website in internet explorer, would check this? IE still is the market leader and a big portion of people will miss your fantastic writing due to this problem.
Woah! I’m really loving the template/theme of this blog. It’s simple, yet effective. A lot of times it’s hard to get that “perfect balance” between usability and visual appearance. I must say that you’ve done a amazing job with this. In addition, the blog loads super quick for me on Opera. Superb Blog!
F*ckin? tremendous issues here. I am very happy to peer your article. Thank you a lot and i’m taking a look ahead to contact you. Will you please drop me a mail?
thanks
Excellent blog you have here.. It’s difficult to find good quality writing like yours nowadays. I seriously appreciate people like you! Take care!!
bookmarked!!, I love your blog.
This website truly has all of the information and facts I wanted concerning this subject and didn’t know who to ask.
Your style is really unique in comparison to other people I’ve read stuff from. Many thanks for posting when you’ve got the opportunity, Guess I’ll just book mark this page.
Superb post however , I was wondering if you could write a litte more on this topic? I’d be very grateful if you could elaborate a little bit further. Kudos!
Thanks for the thoughts you reveal through your blog. In addition, many young women who seem to become pregnant don’t even seek to get health insurance coverage because they worry they won’t qualify. Although a few states currently require that insurers offer coverage no matter the pre-existing conditions. Costs on these types of guaranteed options are usually greater, but when with the high cost of medical care it may be a safer approach to take to protect your financial future.
I am grateful for your post. I would really like to say that the tariff of car insurance will vary from one insurance policy to another, due to the fact there are so many different facets which bring about the overall cost. By way of example, the make and model of the car will have a large bearing on the price. A reliable ancient family car or truck will have a less expensive premium when compared to a flashy fancy car.
Hey there! Do you know if they make any plugins to assist with SEO? I’m trying to get my blog to rank for some targeted keywords but I’m not seeing very good gains. If you know of any please share. Thanks!
naturally like your web site but you have to test the spelling on several of your posts. Many of them are rife with spelling problems and I to find it very bothersome to tell the reality however I will definitely come again again.
Click vào quảng cáo của nó là tự động tải về một đống phần mềm gián điệp.
Click vào quảng cáo của nó là tự động tải về một đống phần mềm gián điệp.
Nhìn chuyên nghiệp vậy thôi chứ bên trong toàn lừa đảo, dụ dỗ đầu tư vớ vẩn.
Hello there, You have done a great job. I?ll definitely digg it and personally recommend to my friends. I’m confident they will be benefited from this website.
Hi colleagues, its fantastic article regarding cultureand entirely explained, keep it up all the time.|
Great post! We will be linking to this particularly great post on our website. Keep up the great writing.
We are a group of volunteers and opening a new scheme in our community. Your site provided us with valuable info to work on. You’ve done a formidable job and our entire community will be thankful to you.
Hi, I do think this is an excellent blog. I stumbledupon it 😉 I’m going to revisit yet again since i have saved as a favorite it. Money and freedom is the greatest way to change, may you be rich and continue to guide other people.
Heya i am for the first time here. I found this board and I find It truly useful & it helped me out a lot. I hope to give something back and help others like you aided me.
okmark your blog and check again here frequently. I’m quite certain I will learn a lot of new stuff right here! Best of luck for the next!
Coi chừng! Trang này cài mã độc theo dõi, vào xong là máy lag như điên.
Bọn này chuyên thu thập thông tin cá nhân rồi đem bán cho tụi đa cấp.
First off I would like to say excellent blog! I had a quick question which I’d like to ask if you do not mind. I was interested to find out how you center yourself and clear your thoughts prior to writing. I’ve had a tough time clearing my thoughts in getting my thoughts out. I truly do take pleasure in writing however it just seems like the first 10 to 15 minutes tend to be lost simply just trying to figure out how to begin. Any recommendations or tips? Thank you!|
Nice post. I learn something more challenging on different blogs everyday. It can all the time be stimulating to learn content material from other writers and practice just a little one thing from their store. I?d favor to make use of some with the content material on my blog whether or not you don?t mind. Natually I?ll offer you a hyperlink on your internet blog. Thanks for sharing.
Thanks for your post. One other thing is the fact that individual states have their particular laws that will affect homeowners, which makes it quite difficult for the our lawmakers to come up with the latest set of recommendations concerning home foreclosure on householders. The problem is that a state possesses own legal guidelines which may work in an undesirable manner in relation to foreclosure procedures.
Its such as you learn my mind! You seem to know a lot approximately this, like you wrote the e-book in it or something. I feel that you simply can do with some percent to pressure the message house a bit, however instead of that, that is fantastic blog. An excellent read. I’ll definitely be back.|
Hey There. I found your blog using msn. This is a really well written article. I will be sure to bookmark it and come back to read more of your useful info. Thanks for the post. I?ll definitely return.
Good post. I learn one thing more difficult on completely different blogs everyday. It’ll always be stimulating to read content material from different writers and observe slightly one thing from their store. I?d choose to make use of some with the content on my weblog whether or not you don?t mind. Natually I?ll provide you with a hyperlink in your internet blog. Thanks for sharing.
Thanks for the recommendations shared using your blog. One more thing I would like to say is that fat loss is not information about going on a celebrity diet and trying to reduce as much weight that you can in a few months. The most effective way in losing weight is by taking it slowly but surely and right after some basic ideas which can provide help to make the most from the attempt to slim down. You may know and be following these tips, however reinforcing know-how never damages.
A few things i have usually told persons is that while searching for a good internet electronics retail store, there are a few components that you have to take into account. First and foremost, you would like to make sure to discover a reputable and reliable retailer that has got great assessments and ratings from other buyers and market sector professionals. This will ensure that you are getting through with a well-known store that provides good assistance and aid to their patrons. Many thanks sharing your notions on this site.
Wow! This can be one particular of the most useful blogs We’ve ever arrive across on this subject. Actually Excellent. I am also an expert in this topic so I can understand your hard work.
What i don’t understood is actually how you are not actually much more well-liked than you might be now. You’re very intelligent. You realize therefore considerably relating to this subject, produced me personally consider it from a lot of varied angles. Its like men and women aren’t fascinated unless it?s one thing to accomplish with Lady gaga! Your own stuffs excellent. Always maintain it up!
Aby utworzyć profil, należy dokonać szybkiej rejestracji, podając adres e-mail i hasło. Wszystkie pozostałe dane o kliencie wypełnia się już po autoryzacji na stronie. Niestety, operator nie przewidział możliwości rejestracji za pośrednictwem popularnych serwisów społecznościowych, co jest powszechnie stosowane na stronach współczesnych polskich kasyn, takich jak Total Casino.
I would like to thank you for the efforts you have put in penning this website. I am hoping to view the same high-grade content from you later on as well. In fact, your creative writing abilities has inspired me to get my own blog now 😉
Find the best ragnarok online private server for you. RateMyRagnarok ranks ragnarok private servers with VPN-protected reviews so ratings stay honest. Browse 40,000+ items, monsters, maps, and skills in our complete ragnarok online database. Compare ragnarok servers side by side, read real player reviews, and discover new ragnarok online servers before anyone else. Whether you’re looking for a ragnarok origin private server, a classic ro private server, or the top ragnarok private servers in 2026, RMR has you covered. List your ragnarok server for free and let the community decide. The RO hub built by players, for players.
Find the best ragnarok online private server for you. RateMyRagnarok ranks ragnarok private servers with VPN-protected reviews so ratings stay honest. Browse 40,000+ items, monsters, maps, and skills in our complete ragnarok online database. Compare ragnarok servers side by side, read real player reviews, and discover new ragnarok online servers before anyone else. Whether you’re looking for a ragnarok origin private server, a classic ro private server, or the top ragnarok private servers in 2026, RMR has you covered. List your ragnarok server for free and let the community decide. The RO hub built by players, for players.
Find the best ragnarok online private server for you. RateMyRagnarok ranks ragnarok private servers with VPN-protected reviews so ratings stay honest. Browse 40,000+ items, monsters, maps, and skills in our complete ragnarok online database. Compare ragnarok servers side by side, read real player reviews, and discover new ragnarok online servers before anyone else. Whether you’re looking for a ragnarok origin private server, a classic ro private server, or the top ragnarok private servers in 2026, RMR has you covered. List your ragnarok server for free and let the community decide. The RO hub built by players, for players.
Find the best ragnarok online private server for you. RateMyRagnarok ranks ragnarok private servers with VPN-protected reviews so ratings stay honest. Browse 40,000+ items, monsters, maps, and skills in our complete ragnarok online database. Compare ragnarok servers side by side, read real player reviews, and discover new ragnarok online servers before anyone else. Whether you’re looking for a ragnarok origin private server, a classic ro private server, or the top ragnarok private servers in 2026, RMR has you covered. List your ragnarok server for free and let the community decide. The RO hub built by players, for players.
Why visitors still make use of to read news papers when in this technological globe all is presented on web?|
For the reason that the admin of this web site is working, no uncertainty very rapidly it will be famous, due to its quality contents.|
Aw, this was an extremely good post. Finding the time and actual effort to produce a very good article… but what can I say… I procrastinate a whole lot and don’t seem to get anything done.
I’m impressed, I have to admit. Rarely do I come across a blog that’s equally educative and entertaining, and without a doubt, you have hit the nail on the head. The problem is something which too few people are speaking intelligently about. I am very happy that I stumbled across this during my search for something concerning this.
This is a topic that is near to my heart… Thank you! Exactly where are your contact details though?
This is a topic which is close to my heart… Thank you! Exactly where are your contact details though?|
I blog often and I truly appreciate your content. The article has really peaked my interest. I am going to bookmark your blog and keep checking for new information about once a week. I opted in for your Feed as well.
This design is wicked! You definitely know how to keep a reader entertained. Between your wit and your videos, I was almost moved to start my own blog (well, almost…HaHa!) Fantastic job. I really loved what you had to say, and more than that, how you presented it. Too cool!|
I simply could not go away your website before suggesting that I really enjoyed the standard information an individual supply for your guests? Is gonna be back steadily to check out new posts|
Thanks for your post. I would also like to say that a health insurance brokerage service also works well with the benefit of the actual coordinators of the group insurance. The health broker is given a directory of benefits desired by a person or a group coordinator. What a broker really does is search for individuals or maybe coordinators which best complement those demands. Then he offers his suggestions and if both parties agree, this broker formulates binding agreement between the two parties.
Thanks for your post. I would really like to comment that the price of car insurance will vary from one plan to another, for the reason that there are so many different facets which play a role in the overall cost. For example, the brand name of the motor vehicle will have an enormous bearing on the fee. A reliable older family car will have a lower priced premium when compared to a flashy sports vehicle.
A few things i have observed in terms of pc memory is the fact that there are technical specs such as SDRAM, DDR or anything else, that must fit the features of the mother board. If the personal computer’s motherboard is fairly current while there are no operating system issues, replacing the memory space literally normally requires under an hour. It’s among the easiest computer system upgrade processes one can visualize. Thanks for revealing your ideas.
I am so happy to read this. This is the type of manual that needs to be given and not the random misinformation that’s at the other blogs. Appreciate your sharing this best doc.
Everything is very open with a very clear explanation of the issues. It was definitely informative. Your website is extremely helpful. Thank you for sharing.
Magnificent beat ! I wish to apprentice whilst you amend your web site, how can i subscribe for a weblog web site? The account aided me a acceptable deal. I had been a little bit acquainted of this your broadcast provided brilliant transparent idea|
I?d must check with you here. Which is not something I often do! I take pleasure in reading a publish that may make folks think. Also, thanks for allowing me to remark!
Hmm it looks like your blog ate my first comment (it was extremely long) so I guess I’ll just sum it up what I submitted and say, I’m thoroughly enjoying your blog. I too am an aspiring blog blogger but I’m still new to everything. Do you have any recommendations for inexperienced blog writers? I’d certainly appreciate it.
GemstoneRO Servers is one of the best Ragnarok Online private servers in 2026, offering two free RO servers: Renewal with EP21 Age of Heroes, chapter 1, chapter2 4th jobs, Doram, latest skills and mid rates, and Loki Pre-Renewal with classic gameplay, custom quests, unique gear and 100x rates. Play on PC or Android with an active global community, low-latency proxies, weekly updates and no paytowin. Join GemstoneRO and enjoy Ragnarok Online your way.
GemstoneRO Servers is one of the best Ragnarok Online private servers in 2026, offering two free RO servers: Renewal with EP21 Age of Heroes, chapter 1, chapter2 4th jobs, Doram, latest skills and mid rates, and Loki Pre-Renewal with classic gameplay, custom quests, unique gear and 100x rates. Play on PC or Android with an active global community, low-latency proxies, weekly updates and no paytowin. Join GemstoneRO and enjoy Ragnarok Online your way.
GemstoneRO Servers is one of the best Ragnarok Online private servers in 2026, offering two free RO servers: Renewal with EP21 Age of Heroes, chapter 1, chapter2 4th jobs, Doram, latest skills and mid rates, and Loki Pre-Renewal with classic gameplay, custom quests, unique gear and 100x rates. Play on PC or Android with an active global community, low-latency proxies, weekly updates and no paytowin. Join GemstoneRO and enjoy Ragnarok Online your way.
GemstoneRO Servers is one of the best Ragnarok Online private servers in 2026, offering two free RO servers: Renewal with EP21 Age of Heroes, chapter 1, chapter2 4th jobs, Doram, latest skills and mid rates, and Loki Pre-Renewal with classic gameplay, custom quests, unique gear and 100x rates. Play on PC or Android with an active global community, low-latency proxies, weekly updates and no paytowin. Join GemstoneRO and enjoy Ragnarok Online your way.
RateMyRagnarok is a modern Ragnarok Online private server ranking and review platform for 2026. Find the best Ragnarok private servers, RO private servers, Renewal servers, Pre-Renewal servers, low-rate, mid-rate, high-rate, custom, mobile, and classic Ragnarok Online servers. Compare rates, features, reviews, uptime, community activity, and server details. List your RO server for free and help players discover trusted Ragnarok Online private servers.
I’m really impressed along with your writing skills and also with the format on your weblog. Is this a paid subject matter or did you customize it your self? Anyway stay up the nice quality writing, it’s uncommon to peer a nice blog like this one these days..|
Hi! I know this is somewhat off topic but I was wondering if you knew where I could find a captcha plugin for my comment form? I’m using the same blog platform as yours and I’m having problems finding one? Thanks a lot!
Can I simply say what a relief to find somebody who really knows what theyre talking about on the internet. You definitely know how you can bring an issue to mild and make it important. Extra individuals must learn this and perceive this side of the story. I cant believe youre no more in style because you definitely have the gift.
I savour, lead to I found just what I was looking for. You’ve ended my 4 day long hunt! God Bless you man. Have a nice day. Bye|
It’s a shame you don’t have a donate button! I’d certainly donate to this fantastic blog! I guess for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to new updates and will share this site with my Facebook group. Talk soon!|
Thanks for your publish. I have continually noticed that a lot of people are wanting to lose weight because they wish to appear slim as well as attractive. Having said that, they do not continually realize that there are more benefits to losing weight also. Doctors say that fat people have problems with a variety of ailments that can be perfectely attributed to their excess weight. The great thing is that people who are overweight in addition to suffering from several diseases can reduce the severity of the illnesses simply by losing weight. It is easy to see a gradual but identifiable improvement in health whenever even a minor amount of losing weight is realized.
I really like what you guys are up too. This kind of clever work and exposure! Keep up the terrific works guys I’ve included you guys to my own blogroll.
I am so happy to read this. This is the type of manual that needs to be given and not the random misinformation that is at the other blogs. Appreciate your sharing this greatest doc.
You made some decent points there. I looked on the internet to find out more about the issue and found most individuals will go along with your views on this site.
Having read this I thought it was rather enlightening. I appreciate you taking the time and energy to put this short article together. I once again find myself spending a lot of time both reading and leaving comments. But so what, it was still worthwhile!
Whats up! I simply wish to give an enormous thumbs up for the great info you will have right here on this post. I will likely be coming back to your blog for extra soon.
This site definitely has all of the information and facts I wanted about this subject and didn’t know who to ask.
This site was… how do you say it? Relevant!! Finally I’ve found something that helped me. Thank you.
It?s arduous to search out knowledgeable people on this topic, but you sound like you realize what you?re speaking about! Thanks
F*ckin? awesome things here. I am very satisfied to peer your article. Thank you a lot and i’m having a look ahead to contact you. Will you kindly drop me a mail?
I do not even know how I finished up right here, but I thought this submit was good. I don’t realize who you might be but certainly you are going to a well-known blogger if you happen to are not already 😉 Cheers!
Wow, this article is mind-blowing! The author has done a phenomenal job of delivering the information in an captivating and educational manner. I can’t thank him enough for providing such precious insights that have undoubtedly enlightened my knowledge in this subject area. Hats off to her for crafting such a masterpiece!
I?ve been exploring for a little for any high quality articles or weblog posts in this kind of space . Exploring in Yahoo I eventually stumbled upon this website. Studying this information So i?m satisfied to convey that I have an incredibly just right uncanny feeling I came upon exactly what I needed. I so much definitely will make sure to do not forget this website and give it a glance regularly.
Terrific work! This is the type of info that should be shared around the web. Shame on the search engines for not positioning this post higher! Come on over and visit my web site . Thanks =)
You made some decent points there. I appeared on the web for the issue and found most people will go together with together with your website.
I am extremely impressed with your writing skills as well as with the layout on your blog. Is this a paid theme or did you customize it yourself? Anyway keep up the nice quality writing, it is rare to see a nice blog like this one nowadays.|
This post is worth everyone’s attention. How can I find out more?|
Aw, this was a very nice post. In thought I want to put in writing like this additionally ? taking time and actual effort to make a very good article? however what can I say? I procrastinate alot and certainly not seem to get something done.
It’s a pity you don’t have a donate button! I’d without a doubt donate to this fantastic blog! I suppose for now i’ll settle for bookmarking and adding your RSS feed to my Google account. I look forward to brand new updates and will talk about this website with my Facebook group. Talk soon!
I’m so happy to read this. This is the kind of manual that needs to be given and not the accidental misinformation that is at the other blogs. Appreciate your sharing this greatest doc.
Very nice write-up. I absolutely appreciate this website. Keep it up!
Superb blog! Do you have any hints for aspiring writers? I’m hoping to start my own website soon but I’m a little lost on everything. Would you propose starting with a free platform like WordPress or go for a paid option? There are so many choices out there that I’m completely confused .. Any tips? Cheers!
Hello there, simply become aware of your weblog thru Google, and found that it is really informative. I’m going to watch out for brussels. I will appreciate for those who continue this in future. Lots of folks will probably be benefited out of your writing. Cheers!|
Hi there! Quick question that’s totally off topic. Do you know how to make your site mobile friendly? My site looks weird when browsing from my iphone 4. I’m trying to find a theme or plugin that might be able to correct this problem. If you have any recommendations, please share. Appreciate it!|
Undeniably believe that which you stated. Your favorite justification appeared to be on the web the simplest thing to be aware of. I say to you, I definitely get annoyed while people think about worries that they just don’t know about. You managed to hit the nail upon the top as well as defined out the whole thing without having side effect , people can take a signal. Will likely be back to get more. Thanks
There’s noticeably a bundle to find out about this. I assume you made sure good factors in features also.
Today, while I was at work, my cousin stole my iPad and tested to see if it can survive a 25 foot drop, just so she can be a youtube sensation. My apple ipad is now destroyed and she has 83 views. I know this is entirely off topic but I had to share it with someone!
Thanks for the concepts you are sharing on this site. Another thing I would really like to say is always that getting hold of some copies of your credit score in order to check accuracy of any detail is the first motion you have to accomplish in fixing credit. You are looking to clean up your credit reports from dangerous details errors that screw up your credit score.
My brother recommended I would possibly like this web site. He used to be totally right. This publish truly made my day. You cann’t believe simply how much time I had spent for this information! Thank you!
What i don’t realize is actually how you’re not really much more well-liked than you might be right now. You’re so intelligent. You realize therefore considerably relating to this subject, made me personally consider it from so many varied angles. Its like women and men aren’t fascinated unless it is one thing to accomplish with Lady gaga! Your own stuffs nice. Always maintain it up!
wonderful publish, very informative. I’m wondering why the opposite experts of this sector do not notice this. You should proceed your writing. I am sure, you have a great readers’ base already!|
Hey! This is kind of off topic but I need some help from an established blog. Is it very difficult to set up your own blog? I’m not very techincal but I can figure things out pretty fast. I’m thinking about making my own but I’m not sure where to begin. Do you have any ideas or suggestions? Thanks
Good day! I simply want to offer you a huge thumbs up for your excellent info you have got here on this post. I will be returning to your site for more soon.
Hey There. I discovered your blog the usage of msn. This is a really neatly written article. I?ll make sure to bookmark it and come back to read extra of your useful info. Thank you for the post. I will certainly return.
I have witnessed that good real estate agents everywhere are Advertising. They are knowing that it’s not only placing a sign in the front place. It’s really concerning building connections with these dealers who sooner or later will become customers. So, whenever you give your time and efforts to helping these sellers go it alone — the “Law of Reciprocity” kicks in. Great blog post.
The things i have observed in terms of laptop memory is always that there are specifications such as SDRAM, DDR and the like, that must go with the specific features of the motherboard. If the personal computer’s motherboard is fairly current while there are no os issues, upgrading the storage space literally normally takes under 1 hour. It’s among the easiest computer upgrade methods one can imagine. Thanks for expressing your ideas.
affordablecanvaspaintings.com.au is Australia Popular Online 100 percent Handmade Art Store. We deliver Budget Handmade Canvas Paintings, Abstract Art, Oil Paintings, Artwork Sale, Acrylic Wall Art Paintings, Custom Art, Oil Portraits, Pet Paintings, Building Paintings etc. 1000+ Designs To Choose From, Highly Experienced Artists team, Up-to 50 percent OFF SALE and FREE Delivery Australia, Sydney, Melbourne, Brisbane, Adelaide, Hobart and all regional areas. We ship worldwide international locations. Order Online Your Handmade Art Today.
affordablecanvaspaintings.com.au is Australia Popular Online 100 percent Handmade Art Store. We deliver Budget Handmade Canvas Paintings, Abstract Art, Oil Paintings, Artwork Sale, Acrylic Wall Art Paintings, Custom Art, Oil Portraits, Pet Paintings, Building Paintings etc. 1000+ Designs To Choose From, Highly Experienced Artists team, Up-to 50 percent OFF SALE and FREE Delivery Australia, Sydney, Melbourne, Brisbane, Adelaide, Hobart and all regional areas. We ship worldwide international locations. Order Online Your Handmade Art Today.
I love it whenever people get together and share thoughts. Great website, stick with it.
Greetings, I think your website may be having web browser compatibility issues. Whenever I take a look at your site in Safari, it looks fine however when opening in I.E., it’s got some overlapping issues. I simply wanted to give you a quick heads up! Besides that, wonderful website!
Your style is unique compared to other people I have read stuff from. Thank you for posting when you’ve got the opportunity, Guess I will just book mark this page.
My brother suggested I might like this website. He was entirely right. This post actually made my day. You can not imagine simply how much time I had spent for this information! Thanks!
What?s Happening i am new to this, I stumbled upon this I have found It positively helpful and it has aided me out loads. I hope to contribute & aid other users like its helped me. Good job.
Spot on with this write-up, I truly believe this amazing site needs a great deal more attention. I’ll probably be returning to read more, thanks for the info.
bookmarked!!, I like your blog!
Very good info. Lucky me I ran across your site by chance (stumbleupon). I have saved it for later.
Your style is unique compared to other people I have read stuff from. Thank you for posting when you’ve got the opportunity, Guess I’ll just book mark this page.
Excellent site you’ve got here.. It’s hard to find excellent writing like yours nowadays. I seriously appreciate individuals like you! Take care!!